Propose a synthesis for each of the following compounds in enantiomerically pure form. Use an asymmetric epoxidation
Question:
Propose a synthesis for each of the following compounds in enantiomerically pure form. Use an asymmetric epoxidation in each synthesis.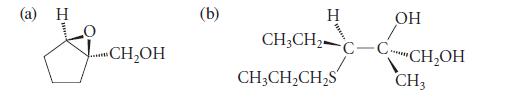
Transcribed Image Text:
(a) H ...CH₂OH (b) H CH3CH₂ CH3CH₂CH₂S C OH CH₂OH CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
3 b H H CHCH DET TiOPr4 CH...View the full answer

Answered By

Chaudhary Deepa Tomar
I graduated with Bachelor of Science in 2015 from CCSU,Meerut. I have certificate in CCC computer language. Now I am pursuing my B.ed from CCSU,Meerut .This education helped further from my analytical mind.My analytical mind of thinking is well suited to teacher role where I oversee students
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis for each of the following compounds in enantiomerically pure form from enantiomerically pure (2R,3R)-2,3-dimethyloxirane: (a) (b) (c) (d) (2R,3S)-3-methoxy-2-butanol O T ||...
-
Outline a synthesis for each of the following compounds in stereochemically pure form from enantiomerically pure (2R,3R)-2,3-dimethyloxirane: (a) (b) CH,O O (3S)-CH CHC CH ., , , OCH (2S,3R)-CH CH...
-
Propose a synthesis for each of the following compounds from butyric acid and any other reagents. (a) 2-methyl-2-pentanol (b) CH3CH2CH2CH2CH2CH2NH2 (c) CH3CH2CH2CH2NH2
-
If the Federal Government increases taxes:What will be the effect on money demand, money supply, and interest rates? Money demand decreases, money supply is unchanged, and interest rates decrease...
-
Kim Lee, the bookkeeper for Briton Company, had never missed a days work for the past 10 years until last week. Since that time, he has not been located. You now suspect that Kim may have embezzled...
-
Each of the following items was found on the financial statements for Edge Company for the year ended December 31, 2011: Income from continuing operations ............... $ 125,000 Gain on the sale...
-
In the organizational chart for the consumer-packaged goods firm in Figure 22 5, where do product line, functional, and geographical groupings occur? Figure 22-5 Chief Marketing Officer or Vice...
-
Pure Decorating uses a job order costing system to collect the costs of its interior decorating business. Each clients consultation is treated as a separate job. Overhead is applied to each job based...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large central IS organization with several operational relational databases at its...
-
Give the major organic product of each of the following reactions. Include stereochemistry where relevant. (a) Dibutyl sulfide with 1 equivalent of H 2 O 2 at 25C (b) Dibutyl sulfide with 2 or more...
-
(a) Use the picture of the catalyst complex in Fig. 11.3a to explain why most E allylic alcohols undergo asymmetric epoxidation more rapidly than their Z isomers. (b) Would the same phenomenon be...
-
How is an airplane able to fly upside down?
-
A city is required to report to the state the mean property tax of its citizens. From previous years, officials know that the population standard deviation is likely to be $975. A 99 % confidence...
-
Suppose the quality control expert of question 30 knows from past experience that the number of defective light bulbs in a case of 10,000 has a population standard deviation of 221. She would like to...
-
Suppose you want to estimate a population proportion p. The total population consists of 1,000, your population consists of 100, and the sample proportion is .42. Find a 95 % confidence interval for...
-
A quality control engineer at the Brite Lite Light Bulb Company wants to test for the number of defective light bulbs in a case of 1,200. From past experience, he knows that the population standard...
-
The president of Prof. Andersons university is interested in the proportion of students favoring the new campus bus system. (a) Since there are 13,200 students and the president would like to have a...
-
How is capital gain income treated differently from other forms of income?
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Proteins can be cleaved specifically at the amide bond on the carboxyl side of methionine residues by reaction with cyanogen bromide, BrC = N. The reaction occurs in several steps: (a) The first step...
-
A clever new method of peptide synthesis involves formation of an amide bond by reaction of an -keto acid with an N-alkyl hydroxylamine: The reaction is thought to occur by nucleophilic addition of...
-
Arginine, the most basic of the 20 common amino acids, contains a guanidino functional group in its side chain. Explain, using resonance structures to show how the protonated guanidino group...
-
Major software companies, after years of providing unlimited free telephone technical support for their products, have recently begun to charge for these services ( typically after an initial start -...
-
Company A carries 5000 items. Customers ordered 1000 items from inventory last week. Company A was able to fulfill 600 items of the order. What is the in-stock probability for last week?
-
Company B sold 50% of the 5,000 items that it carried last week. There were 500 items for which some demand was not satisfied. What is the stockout probability for last week?

Study smarter with the SolutionInn App


