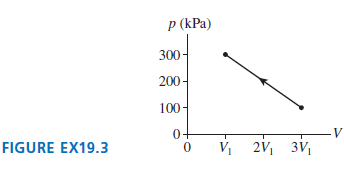
80 J of work are done on the gas in the process shown in Figure EX19.3. What
Question:
Transcribed Image Text:
p (kPa) 300- 200- 100- VI 2V1 FIGURE EX19.3 3V1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
Model Assume the gas is ideal The work done on a gas is the negative of the area under the ...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Three different processes act on a system. (a) In process A, 42 J of work are done on the system and 77 J of heat are added to the system. Find the change in the system's internal energy. (b) In...
-
500 J of work are done on a system in a process that decreases the systems thermal energy by 200 J. How much heat energy is transferred to or from the system?
-
Juana slides a crate along the floor of the moving van. The coefficient of kinetic friction between the crate and the van floor is 0.120. The crate has a mass of 56.8 kg and Juana pushes with a...
-
Bloomberg Intelligence listed 50 companies to watch in 2018 (www .bloomberg.com/features/companies-to-watch-2018). Twelve of the companies are listed here with their total assets and 12-month sales....
-
The neutrons in a parallel beam, each having kinetic energy 0.025 eV, are directed through two slits 0.40 mm apart. How far apart will the interference peaks be on a screen 1.0 m away?
-
(a) What is treasury stock? (b) Why might a corporation acquire treasury stock?
-
What form of representation would you recommend for this new market or would you consider setting up a manufacturing subsidiary? Give reasons for your decision.
-
Under U. S. GAAP, property, plant, and equipment are reported at historical cost net of accumulated depreciation. These assets are written down to fair value when it is determined that they have been...
-
A proton, moving with a velocity of v, collides elastically with another proton that is initially at rest. Assuming that after the collision the speed of the initially moving proton is 3.00 times the...
-
The Following Items/Events Relate To Hospitals Covered By The AICPA Audit Guide On Health Care Providers And FAS #116, #117, #124, And #136. Each Choice May Be Used More Than Once. Classify Each Of...
-
Do (a) Temperature, (b) Heat, (c) Thermal energy describe a property of a system, an interaction of the system with its environment, or both? Explain.
-
Two containers hold equal masses of nitrogen gas at equal temperatures. You supply 10 J of heat to container A while not allowing its volume to change, and you supply 10 J of heat to container B...
-
Langley uses a job costing system. At the beginning of June, two orders were in process as follows: There was no inventory in finished goods on 1 June. During June, orders numbered 106 to 120 were...
-
Store the values in a hash table with 20 positions, using the division method of hashing and the linear probing method of resolving collisions. 66 47 87 90 126 140 145 153 177 285 393 395 467 566 620...
-
This text book follows the common practice of describing indirect costs as overhead costs. The following extract from the NHS costing manual gives separate definitions to indirect costs and overhead...
-
What is meant by the statement Programmer time is an efficiency consideration? Give an example of a situation in which programmer time is used to justify the choice of an algorithm (possibly at the...
-
In Chapter 6, we discussed how to store a linked list in an array of nodes using index values as pointers and managing our list of free nodes. We can use these same techniques to store the nodes of a...
-
Determine the Big-O measure for QuickSort based on the number of elements moved rather than the number of comparisons 1. for the best case. 2. for the worst case.
-
Explain when a unilateral mistake can lead to rendering a contract voidable.
-
Problem 2. (0.6 points, 0.2 points for each question) (a) A company turns its inventory 2 times a month. Its months-of-supply = Its days-of-supply = Please show your analysis below: _months. days. (1...
-
Iodine-131, a beta emitter, has a half-life of 8 days. A 2-gram sample of initially pure iodine-131 is stored for 32 days. How much iodine-131 remains in the sample afterward?
-
The amount of carbon-14 in an ancient wooden bowl is found to be one-half that in a new piece of wood. How old is the bowl?
-
A nucleus of element 112coperniciumis formed using the reaction equation given near the end of Section 11.7. It then undergoes six successive alpha decays. Give the identity of the isotope that...
-
Warnerwoods Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for March. Submit Date March 1 March 5 March 9 March 18 March 25 March 29...
-
Wilmington Company has two manufacturing departments-Assembly and Fabrication. It considers all of its manufacturing overhead costs to be fixed costs. The first set of data that is shown below is...
-
What are the fundamental differences between preemptive and cooperative multitasking in an operating system, and in which scenarios would each be most effective ?

Study smarter with the SolutionInn App


