Question: Estimate the electric fields E 1 and E 2 at points 1 and 2 in Figure Q26.4. Dont forget that E is a vector. ov
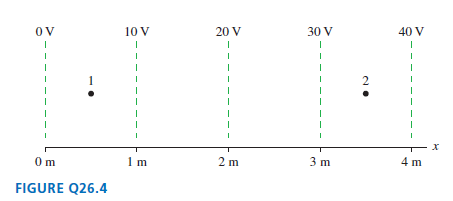
ov OV 10 V 20 V 30 V 40 V 2 m 0m 4 m FIGURE Q26.4
Step by Step Solution
3.26 Rating (164 Votes )
There are 3 Steps involved in it
Both fields point left in t... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
1442_6054778bb8c98_694338.pdf
180 KBs PDF File
1442_6054778bb8c98_694338.docx
120 KBs Word File


