Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured
Question:
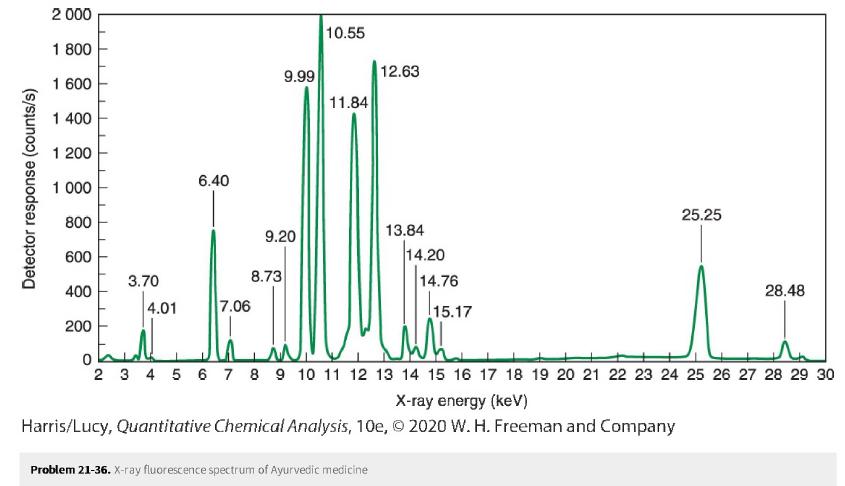
Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several toxic elements. In the spectrum below, identify as many peaks as you can in the X-ray fluorescence spectrum of the Ayurvedic medicine.
Transcribed Image Text:
Detector response (counts/s) 2 000 1 800 1 600 1 400 1 200 1 000 800 600 400 200 3.70 4.01 6.40 7.06 9.99 9.20 8.73 10.55 11.84 12.63 13.84 Problem 21-36. X-ray fluorescence spectrum of Ayurvedic medicine 14.20 14.76 15.17 X-ray energy (keV) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company 25.25 0 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 28.48
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Answer The peaks in the Xray fluorescence spectrum are 1 Cadmi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Holt Industries, Inc. does business in State 1. That state uses federal taxable income as the starting point for calculating state income tax. Interest on state obligations is taxed, but interest on...
-
1. Comment (briefly) on the design of the two costing systems + draw an overview diagram of each system. Explain why you believe / do not believe that the ABC system provides better costing...
-
Ayurveda is a form of medicine practiced in India. In one study, 20% of U.S.- and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several...
-
ABC Pty Ltd would like to set up a Virtualisation Platform on their organisation. You have been hired by Company to be their network and system administrator to implement virtualisation for...
-
Show that is as given in Eq. (11.2.5).
-
The following information is from the 20X1 annual report of Weber Corporation, a company that supplies manufactured parts to the household appliance industry. Average total assets $24,500,000 Average...
-
Using only the factor formulas given in Table 2.6, derive Equation 7.5 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
Linda Butler is the new division controller of the snack-foods division of Daniel Foods. Daniel Foods has reported a minimum 15% growth in annual earnings for each of the past 5 years. The...
-
why is cardinality important to a relational database model? How is it usually defined and what types of GIS analysis operations is it particularly important to? Provide specific examples in your...
-
Anita and her family moved, and her parents feel Anita is not adjusting well to the move. Anita wants to return to her old school to be with her friends. Her parents ask the worker to talk to Anita...
-
Signal in attenuated total reflectance is related to penetration depth. a. Calculate the penetration depth of 1 500 cm -1 1500 cm -1 radiation into an organic sample (n2=1.5)(n 2 =1.5) for a 45450...
-
a. Explain the purpose of the collision cell in Figure 21-28. b. Determination of 28Si 28Si , < experiences isobaric interference from 14N2+ 14N + 2 and background silicon leached from glass...
-
Space Infonautics, Inc., produces handheld Windows CE-compatible organizers. Space Infonautics markets three different handheld models: MobilePro is a souped-up version for the executive on the go,...
-
HELP ME NEED AN IDEA!!!! JUST A PARAGRAPGH Lesson 3 Discussion: Declaring the Brand For Your Strategy Document 2323 unread replies.2323 replies. As mentioned in the audit assignment, you will be...
-
Your firm is contemplating the purchase of a new $642,000 computer-based order entry system. The system will be depreciated straight-line to zero over its six-year life. It will be worth $46,000 at...
-
Use the case knowledge to justify the importance of the work-life balance from both the employer's and the employee's viewpoints. Can you advise Aamir on some industry best policies and practices...
-
help me with UML Diagram, i am a bit lost the code as follows below //Java Code:- package billing; import java.io.File; import java.io.FileWriter; import java.util.ArrayList; import...
-
The sales of mouthwash at MARKAL Pharmacy over the past 6 months have averaged 2,000 cases per month, which is the current order quantity. Markal's cost is $12.00 per case. The company estimates its...
-
Refrigerant-134a is compressed by a compressor from the saturated vapor state at 0.14 MPa to 0.9 MPa and 608C at a rate of 0.108 kg/s. The refrigerant is cooled at a rate of 1.10 kJ/s during...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
The cumulative formation constant for SnCl2(aq) in 1.0 M NaNO3 is 2 = 12. Find the concentration of SnCl2(aq) for a solution in which the concentrations of Sn2+ and Cl- are both somehow fixed at 0.20...
-
Given the following equilibria, calculate the concentration of each zinc species in a solution saturated with Zn(OH)2(s) and containing [OH-] at a fixed concentration of 3.2 10-7M. Zn(OH)2(s) Ksp =...
-
Although KOH, RbOH, and CsOH have little association between metal and hydroxide in aqueous solution, Li+ and Na+ doform complexes with OH-: Na + + OH- NaoH (aq) K1 = 0.20 Prepare a table like the...
-
3. Using the force-time graphs rank the materials from most safe to least safe. (Use 1 for most safe and 4 for the least safe) Force (N) 20 Force (N) Polyurethane Foam 20 Time (sec) Styrofoam 10 Time...
-
Calculate the gravitational force of attraction for two protons separated by 88.0 pm. The mass of a proton is mp = 1.673 10-27 kg. Express your answer using two significant figures. Fg Submit Request...
-
1. (10 points) Assume a 64 entry write-through, direct mapped cache with 4 words per block. The system uses 32-bit addresses and a data word consists of 2-bytes, answer the questions that follow: (a)...

Study smarter with the SolutionInn App


