(a) The companys chemical development group completed a three-step synthesis (outlined below) of compound E from which...
Question:
(a) The company’s chemical development group completed a three-step synthesis (outlined below) of compound E from which they proposed to make molecules of the type D.
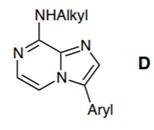
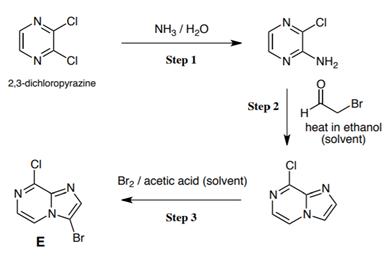
I. Outline a mechanism for Step
II. Account for the selectivity seen in the electrophilic aromatic substitution reaction that has occurred in Step 3. Give a brief mechanistic justification of your answer. 5 marks
(b) Treatment of compound E with 1-hexylamine resulted in the exclusive displacement of the chloro group at position 7 to give compound F.
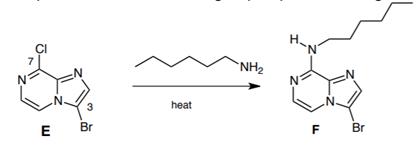
I. Give a brief mechanistic justification of why reaction did not also occur at position 3 to displace the bromo group in an analogous process. 4 marks
II. Three of the four nitrogens atoms of compound F are basic but the fourth is not protonated, even under very acidic conditions. Identify this non-basic nitrogen and explain why it is unreactive. 2 marks

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder




