Question: Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar linear tetrahedral H2 tetrahedral bent tetrahedral H2 trigonal
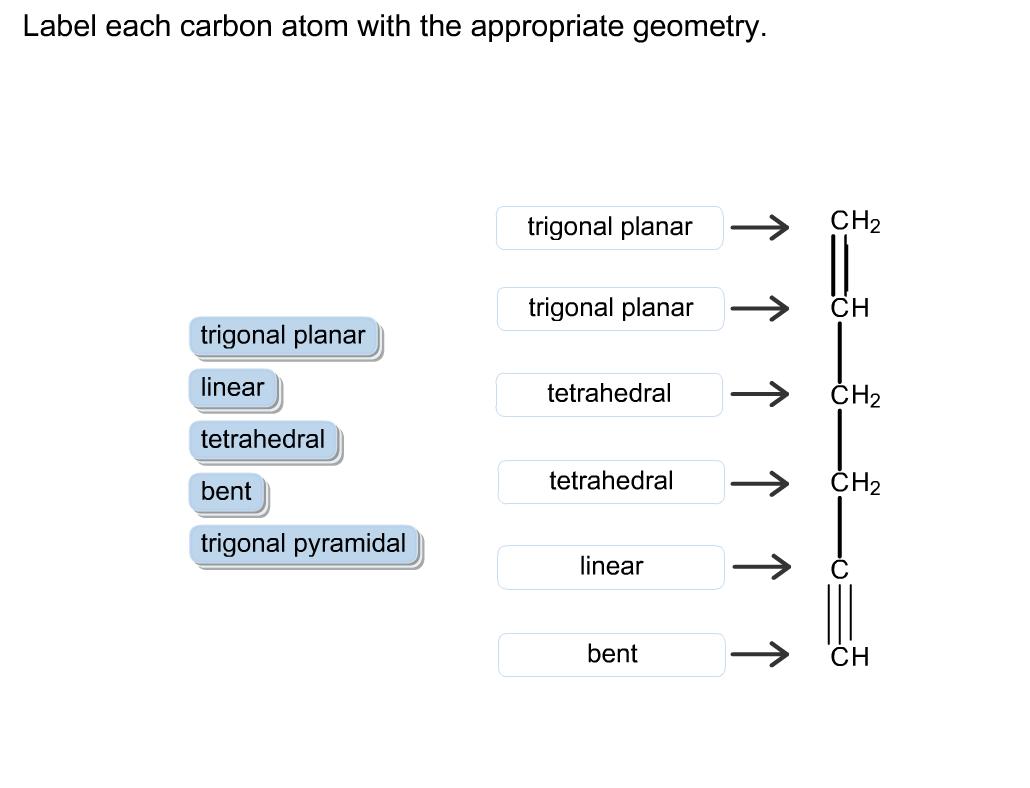
Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar linear tetrahedral H2 tetrahedral bent tetrahedral H2 trigonal pyramidal linear bent CH
Step by Step Solution
3.44 Rating (157 Votes )
There are 3 Steps involved in it
In general Hybridisation Geometry Sp3 ... View full answer

Get step-by-step solutions from verified subject matter experts


