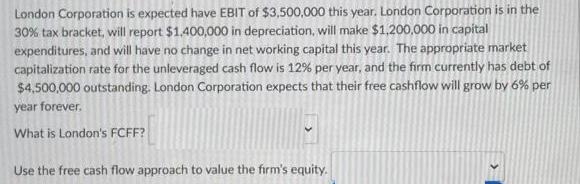
London Corporation is expected have EBIT of $3,500,000 this year. London Corporation is in the 30%...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
London Corporation is expected have EBIT of $3,500,000 this year. London Corporation is in the 30% tax bracket, will report $1,400,000 in depreciation, will make $1,200,000 in capital expenditures, and will have no change in net working capital this year. The appropriate market capitalization rate for the unleveraged cash flow is 12% per year, and the firm currently has debt of $4,500,000 outstanding. London Corporation expects that their free cashflow will grow by 6% per year forever. What is London's FCFF? Use the free cash flow approach to value the firm's equity. London Corporation is expected have EBIT of $3,500,000 this year. London Corporation is in the 30% tax bracket, will report $1,400,000 in depreciation, will make $1,200,000 in capital expenditures, and will have no change in net working capital this year. The appropriate market capitalization rate for the unleveraged cash flow is 12% per year, and the firm currently has debt of $4,500,000 outstanding. London Corporation expects that their free cashflow will grow by 6% per year forever. What is London's FCFF? Use the free cash flow approach to value the firm's equity.
Expert Answer:
Answer rating: 100% (QA)
Calculate London Corporations Free Cash Flow to the Firm FCFF FCFF EBIT1 Tax Rate Depr... View the full answer

Related Book For 

Financial Reporting Financial Statement Analysis and Valuation a strategic perspective
ISBN: 978-1337614689
9th edition
Authors: James M. Wahlen, Stephen P. Baginski, Mark Bradshaw
Posted Date:
Students also viewed these finance questions
-
This case study on project evaluation is applicable for beginning courses in corporate finance or finance strategy. Two alternative investment options are available to evaluate. Challenges are...
-
The MoMi Corporations income before interest, depreciation and taxes, was $1.7 million in the year just ended, and it expects that this will grow by 5% per year forever. To make this happen, the firm...
-
The MoMi Corporations cash flow from operations before interest and taxes was $2 million in the year just ended, and it expects that this will grow by 5% per year forever. To make this happen, the...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
A piston/cylinder in a car contains 12 in 3 of air at 13 lbf/in 2, 68 F. The air is compressed in a quasi-equilibrium polytropic process with polytropic exponent n 1.25 to a final...
-
Actuarial Gains and Losses: The following information relates to the pension plan of CCL Corporation, which has a contributory defined benefit pension plan: The following cases are independent. Case...
-
Following are observed frequencies. The null hypothesis is H0: p1 = 0.5, p2 = 0.3, p3 = 0.15, p4 = 0.05. a. Compute the expected frequencies. b. Compute the value of X2. c. How many degrees of...
-
On January 31, the managers of Integra, Inc., seek to determine the cost of producing their product during January for product pricing and control purposes. The company can easily determine the costs...
-
In our first three lectures, we were focused on the four core principles of economics that form the foundation of the entire course. The goal is not to memorize these principles; it is to train our...
-
Sales of vegetable dehydrators at Bud Baniss discount department store in Gander over the past year are shown below. Management prepared a forecast using a combination of exponential smoothing and...
-
Marlene has a life insurance policy with ABC Insurance. The policy has a 30-day grace period and a death benefit of $250,000. Marlene's husband, Marcus, is the beneficiary on the policy. Marlene's...
-
Using the information in the reading titled 'Cash Flow Projections in Lesson 4 to answer the following question in the space provided. Courtney Fyfe owner of a new hat importing business "Fyfe...
-
Greg Brady formed Johnny Bravo, Inc. (JBI) on January 1, 20x1 by contributing $100,000 in exchange for 100% of JBI stock. JBI immediately (and properly) elected S corporation status. During 20x1, JBI...
-
JOSEI IS TESTIFYING AT THE EMBEZZLEMENT TRIAL OF MARTHA.. SHE TESTIFIES THAT SHE KNEW MARTHA WAS STEALING AND ON OCCASSION HAD INTENTIONALLY HELPED TO COVER THE THEFTS BECAUSE SHE, TOO, HATED THE...
-
The thesis statement explains the position the paper will take in analyzing the topic. The outline provides you with the framework and an organizational structure for you to write a logical and...
-
Based on the below graph, can the Malthusian growth model explain the evolution of population and income per capita in the world up to 1750? Explain GDP Per Capits 6000 5000 4000 3000 2000 1000 500...
-
Prepare the cost of sales section and supply the missing amounts in each of the following: A B D 2,480 14,000 6,200 35,000 77,500 ? 5,000 12,000 19,000 Purchase Returns & Allowances Sales Returns &...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
What does market efficiency mean? What does market efficiency not mean? Explain how market efficiency relates to the amount of information that affects share prices and the speed with which...
-
Problem 13.18 and Exhibit 13.6 in Chapter 13 present selected hypothetical data from projected financial statements for Steak 'n Shake for Year +1 to Year +11. The amounts for Year +11 reflect a...
-
What is a LIFO layer liquidation? How does it affect the prediction of future earnings?
-
Hydrogen gas is expanded reversibly and adiabatically from a volume of \(2.12 \mathrm{dm}^{3}\) at a pressure of \(4 \mathrm{~atm}\) and \(32^{\circ} \mathrm{C}\) until the volume is doubled....
-
If \(C_{P}=a+b T+C T^{2}\), derive a relation to the isobaric mean heat capacity \(\dot{Q}\).
-
A spherical balloon of 1 m diameter contains a gas at 120 kPa. The gas inside the balloon is heated until the pressure reaches 360 kPa. During heating the pressure of the gas inside the balloon is...

Study smarter with the SolutionInn App


