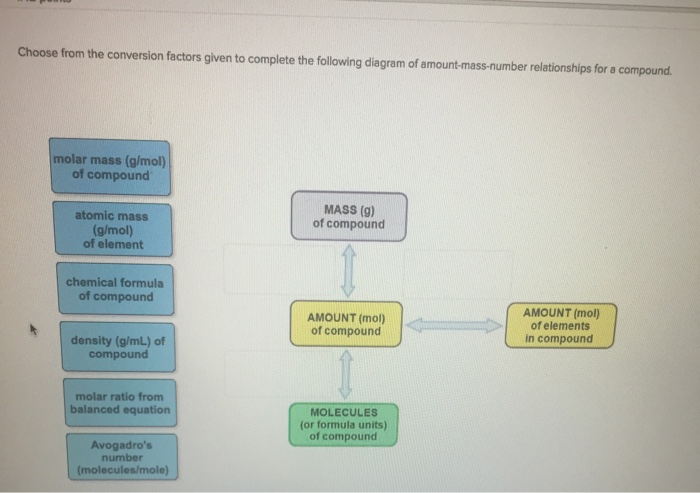
Choose from the conversion factors given to complete the following diagram of amount-mass-number relationships for a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
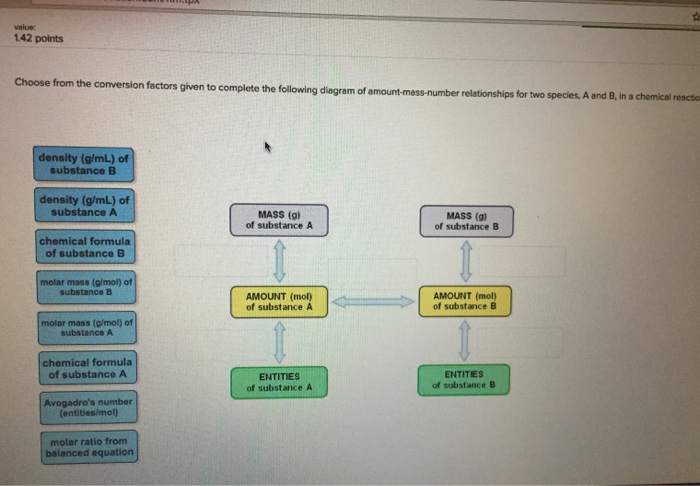
Choose from the conversion factors given to complete the following diagram of amount-mass-number relationships for a compound. molar mass (g/mol) of compound atomic mass (g/mol) of element chemical formula of compound density (g/mL) of compound molar ratio from balanced equation Avogadro's number (molecules/mole) MASS (g) of compound AMOUNT (mol) of compound MOLECULES (or formula units) of compound AMOUNT (mol) of elements in compound value: 142 points Choose from the conversion factors given to complete the following diagram of amount-mass-number relationships for two species, A and B, in a chemical reaction density (g/mL) of substance B density (g/mL) of substance A chemical formula of substance B molar mass (g/mol) of substance B molar mass (g/mol) of substance A chemical formula of substance A Avogadro's number. (entities/mol) molar ratio from balanced equation MASS (g) of substance A AMOUNT (mol) of substance A ENTITIES of substance A MASS (g) of substance B AMOUNT (mol) of substance B ENTITIES of substance B Choose from the conversion factors given to complete the following diagram of amount-mass-number relationships for a compound. molar mass (g/mol) of compound atomic mass (g/mol) of element chemical formula of compound density (g/mL) of compound molar ratio from balanced equation Avogadro's number (molecules/mole) MASS (g) of compound AMOUNT (mol) of compound MOLECULES (or formula units) of compound AMOUNT (mol) of elements in compound value: 142 points Choose from the conversion factors given to complete the following diagram of amount-mass-number relationships for two species, A and B, in a chemical reaction density (g/mL) of substance B density (g/mL) of substance A chemical formula of substance B molar mass (g/mol) of substance B molar mass (g/mol) of substance A chemical formula of substance A Avogadro's number. (entities/mol) molar ratio from balanced equation MASS (g) of substance A AMOUNT (mol) of substance A ENTITIES of substance A MASS (g) of substance B AMOUNT (mol) of substance B ENTITIES of substance B
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
please choose the correct answer 3) The form used to determine an employee's net claim code is called a * A) T4. B) Record of Employment. C) TD1. D) Employee Earnings Record. 4) Income tax deduction...
-
Please choose one of these ten business case studies: A Sweet Breakfast Memory That Connects With the Wrong Market Seeking Even Faster Growth, an E-Commerce Company Stumbles A Business Owner Seeks an...
-
Choose an organization in your community. What questions would you ask to complete an audit of its social responsibility practices?
-
North Folk Dairy has an unused bank revolving credit of $50 million. North Folk Dairys vendor provided an invoice in the amount of $100,000 with payment terms of 2/10, net 45. Compute the nominal...
-
Find online the annual 10-K report for Peets Coffee and Tea (PEET) for fiscal year 2011 (filed in January 2012). Answer the following questions from the notes to their financial statements: a. What...
-
Give the metric symbol, or abbreviation, for each prefix. 1. Hecto 2. Kilo 3. Milli 4. Deci 5. Mega 6. Deka 7. Centi 8. Micro
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
Interact Systems, Inc., has developed software tools that help hotel chains solve application integration problems. Interacts application integration server (AIS) provides a two-way interface between...
-
My elevator pitch for the purchase of an MRI machine is as follows: Hi there, my name is Kayla and I am one of the administrators here at Highlands Regional. Did you know that our MRI machine was...
-
Ivanhoe Machinery Corporation, a private company following ASPE sold manufacturing equipment for $2,500 each. Each machine carried with it a 2-year warranty against manufacturing defects. From...
-
If you are asked to develop the executive compensation plans for TD Bank or Tim Hortons, how will you structure executive compensation in terms of key components, compensation levels and mix? How do...
-
Part A A stone is suspended from the free end of a wire that is wrapped around the outer rim of a pulley, similar to what is shown in (Figure 1). The pulley is a uniform disk with mass 10.0 kg and...
-
What are cultural factors that influence leadership and contribute to global leadership effectiveness.
-
To adjust the incorrect entry, I recommend recognizing $50,000 of the unearned revenue as earned revenue at the end of December. This is because the company has provided one month's worth of...
-
15.13 Programming Assignment #12 Various websites like Wikipedia or IMDB list not just a person's birthdate but also the person's current age. Given a person's birthdate and current date, output the...
-
During the year, Mickey worked as a driver. He is single and has no other sources of income. His qualified business income from driving is $48,000. Assume that Mickey takes the standard deduction of...
-
In 2009, the average price of a new home sold in a country was $294,000. In 2004, the average price of a new home in the same country was $297,000. Let y be the average price of a new home in the...
-
A copper wire (density = 8.96 g/cm 3 ) has a diameter of 0.25 mm. If a sample of this copper wire has a mass of 22 g, how long is the wire?
-
How many bonds could each of the following chelating ligands form with a metal ion? a. Acetylacetone b. Diethylene triamine c. Salen d. Porphine CH2-C-CH NH2-CH-CH-NH-CH2-CH2-NH2 OH HO NH N N HN
-
What is the difference between core electrons and valence electrons? Why do we emphasize the valence electrons in an atom when discussing atomic properties? What is the relationship between valence...
-
A gas contains a mixture of NH 3 (g) and N 2 H 4 (g), both of which react with O 2 (g) to form NO 2 (g) and H 2 O(g). The gaseous mixture (with an initial mass of 61.00 g) is reacted with 10.00 moles...
-
The sequence in Figure P2.2 represents a ball rolling into a wall and bouncing off of it. The ball is \(10 \mathrm{~mm}\) in diameter. Make a graph showing the distance from the leading edge of the...
-
You walk \(3.2 \mathrm{~km}\) to the supermarket and then back home. What is your distance traveled? What is your displacement?
-
Your class observed several different objects in motion along different lines. Figure P2.5 shows some of the graphs other students made of the events. They have labeled the horizontal axis "time" and...

Study smarter with the SolutionInn App


