Steam enters the high-pressure turbine of a steam power plant that operates on the ideal reheat...
Fantastic news! We've Found the answer you've been seeking!
Question:
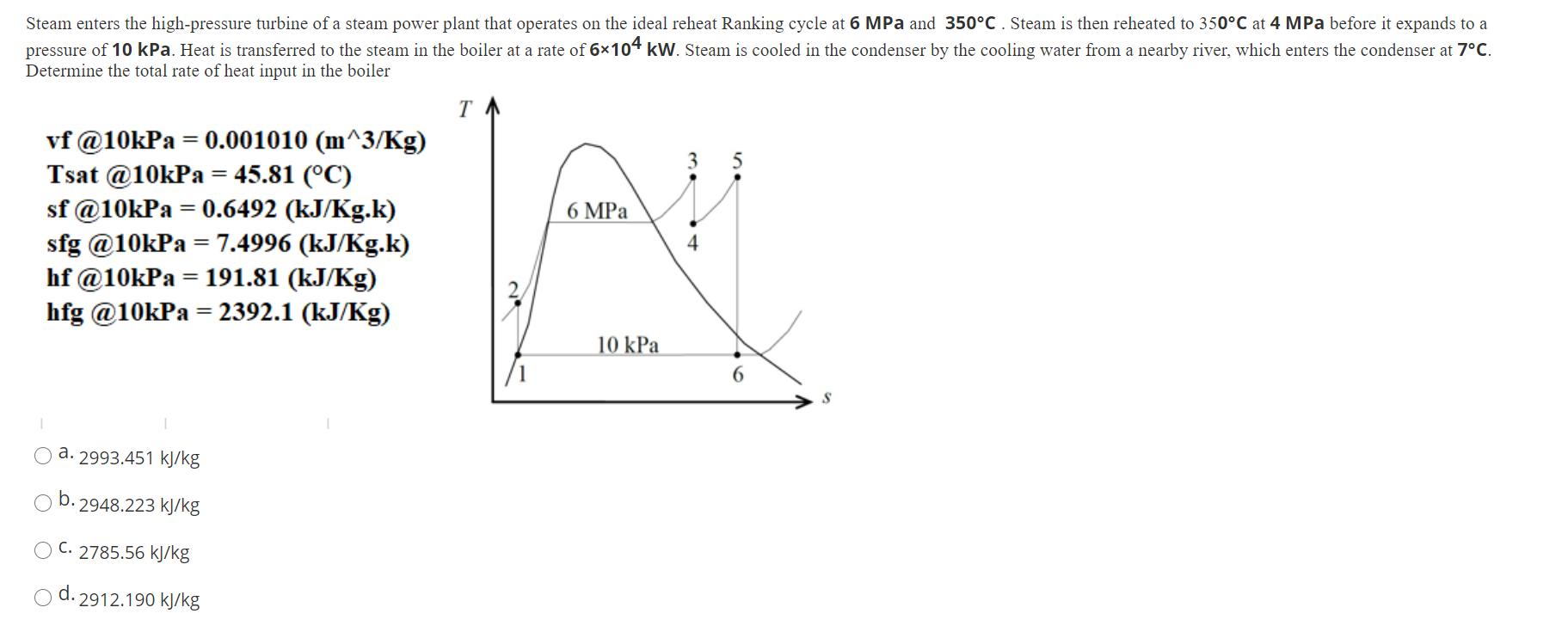
Transcribed Image Text:
Steam enters the high-pressure turbine of a steam power plant that operates on the ideal reheat Ranking cycle at 6 MPa and 350C. Steam is then reheated to 350C at 4 MPa before it expands to a pressure of 10 kPa. Heat is transferred to the steam in the boiler at a rate of 6104 kW. Steam is cooled in the condenser by the cooling water from a nearby river, which enters the condenser at 7C. Determine the total rate of heat input in the boiler vf @10kPa = 0.001010 (m^3/kg) Tsat @10kPa = 45.81 (C) sf @10kPa = 0.6492 (kJ/Kg.k) sfg @10kPa = 7.4996 (kJ/Kg.k) hf @10kPa = 191.81 (kJ/Kg) hfg @10kPa = 2392.1 (kJ/Kg) a. 2993.451 kJ/kg b. 2948.223 kJ/kg OC. 2785.56 kJ/kg d. 2912.190 kJ/kg T /1 6 MPa 10 kPa 4 5 6 Steam enters the high-pressure turbine of a steam power plant that operates on the ideal reheat Ranking cycle at 6 MPa and 350C. Steam is then reheated to 350C at 4 MPa before it expands to a pressure of 10 kPa. Heat is transferred to the steam in the boiler at a rate of 6104 kW. Steam is cooled in the condenser by the cooling water from a nearby river, which enters the condenser at 7C. Determine the total rate of heat input in the boiler vf @10kPa = 0.001010 (m^3/kg) Tsat @10kPa = 45.81 (C) sf @10kPa = 0.6492 (kJ/Kg.k) sfg @10kPa = 7.4996 (kJ/Kg.k) hf @10kPa = 191.81 (kJ/Kg) hfg @10kPa = 2392.1 (kJ/Kg) a. 2993.451 kJ/kg b. 2948.223 kJ/kg OC. 2785.56 kJ/kg d. 2912.190 kJ/kg T /1 6 MPa 10 kPa 4 5 6
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
Steam enters the high-pressure turbine of a steam power plant that operates on the ideal reheat Rankine cycle at 800 psia and 900F and leaves as saturated vapor. Steam is then reheated to 800F before...
-
Steam enters the turbine of a steam power plant that operates on a simple ideal Rankine cycle at a pressure of 6 MPa, and it leaves as a saturated vapor at 7.5 kPa. Heat is transferred to the steam...
-
Consider a steam power plant that operates on the ideal reheat Rankine cycle. The plant maintains the boiler at 5000 kPa, the reheat section at 1200 kPa, and the condenser at 20 kPa. The mixture...
-
Find a PTLU factorization of the given matrix A. (Enter each matrix in the form [[row 1], [row 2],...], where each row is a comma-separated list.) 0 1 2 -1 4 1 1 3 3 A =
-
What is LIBOR? Why is it important?
-
Regardless of the risk management approach chosen, effective risk governance for an investment includes tax risk management. Taxes can effectively impair a trade or investment return if high, and...
-
I want sales representatives who can stand on their own. Once they have been through training and show how to apply their knowledge, it shouldnt be necessary for me to constantly tell them how they...
-
Vidi Corporation made the following purchases related to its property, plant, and equipment during its fiscal year ended December 31, 2014. The company uses the straight-line method of depreciation...
-
My question is: The article uses the term 'self-efficacy' several times. Briefly describe what you understand by self-efficacy in the context of the paper and whether you think it has been clearly...
-
1. Liam Richardson is the business manager for the Smith & Lyngate Insurance agencies in the state of Maryland. Liam is interested in increasing the number of agents in Baltimore and plans to buy...
-
Nancy (53) is a casual smoker, divorced, and is a successful sales representative for a multi-national, over-the-counter health-care products manufacturer. She operates by contract and is...
-
ACTIVITY 2 The letters A to H in the table below represent eight organic compounds. CH, HH A 2.1 C-CH-Br CH- CH CH CH CH CH-CH-CH-C CH3-CH2-CH-C H HIC-H H CH3 H OH CH C-CECICL H Write down the IUPAC...
-
4. (3 pts) Which numbered hydrogen has the lowest pKa? Explain your answer in words and by using structures. 4 1 H H- H SP3 IN H 2 SP 3 I w H Spa Sp3 HH 5
-
Colligative Properties (ALEKS lab) Introduction Lab Data Verify your mass calculation Verify your molality calculation Did you account for the freezing-point of your DI water? Did you report your...
-
( a ) Which two ( out of the three ) would be the best lead compounds? Why? ( b ) Describe the differences between the affinities and efficacies of the two compounds you selected in ( a ) . 4. 100%...
-
A solution of methyl acetate (C3HO) and water is boiling at 99.5 C. A sample of the vapor above the solution is cooled until it condenses. This condensed sample is analyzed, and turns out to be 10.%...
-
At the beginning of the year 2003, accrued liabilities of a pension plan for the employees of AAA Life Insurance Company were 100,000. The assets of the plan at the same time were 150,000. On January...
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
A piston-cylinder device contains an ideal-gas mixture of 3 kmol of He gas and 7 kmol of Ar gas at 50oC and 400 kPa. Now the gas expands at constant pressure until its volume doubles. The amount of...
-
Express Amagat's law of additive volumes. Does this law hold exactly for ideal-gas mixtures? How about nonideal-gas mixtures?
-
In a combustion chamber, ethane (C2H6) is burned at a rate of 8 kg/h with air that enters the combustion chamber at a rate of 176 kg/h. Determine the percentage of excess air used during this process.
-
Verify that the log-likelihood of model (7.7) is \(\sum_{i=1}^{k}\left[n_{i} \lambda-\exp (\lambda) ight]\). (a) Compute MLE of \(\lambda\). (b) Compute the Pearson chi-square statistic and compare...
-
Think about the general concept of a relationship, not necessarily in a business setting, but just relationships in general between any two parties. What aspects of relationships are inherently...
-
What is value? In what ways does a relationship selling approach add value to your customers, to you the salesperson, and to your sales organization?

Study smarter with the SolutionInn App



