The decomposition reaction 2NO, (g)^2NO(g) + O(g) is started in a closed cylinder under isothermal isochoric...
Fantastic news! We've Found the answer you've been seeking!
Question:
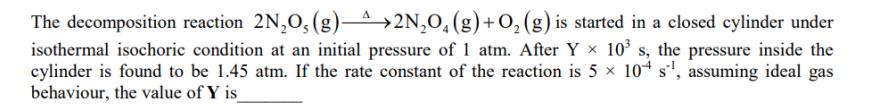
Transcribed Image Text:
The decomposition reaction 2NO, (g)^2NO(g) + O(g) is started in a closed cylinder under isothermal isochoric condition at an initial pressure of 1 atm. After Yx 10 s, the pressure inside the cylinder is found to be 1.45 atm. If the rate constant of the reaction is 5 10 s, assuming ideal gas behaviour, the value of Y is_ The decomposition reaction 2NO, (g)^2NO(g) + O(g) is started in a closed cylinder under isothermal isochoric condition at an initial pressure of 1 atm. After Yx 10 s, the pressure inside the cylinder is found to be 1.45 atm. If the rate constant of the reaction is 5 10 s, assuming ideal gas behaviour, the value of Y is_
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A gas has an initial pressure of 87.0 torr and an initial volume of 28.5 mL. What is its new volume if pressure is changed to 206 torr? Assume temperature and amount are held constant.
-
A gas has an initial pressure of 633 torr and an initial volume of 87.3 mL. What is its new pressure if volume is changed to 45.0 mL? Assume temperature and amount are held constant.
-
1 (one) kg of air at an initial pressure of 8 bar and 100 C is expanded polytropically with n = 1,2 so that the pressure drops as much as one-fifth of the initial pressure. Determine: a. Specific...
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
What problems are occurring with the training sessions? How can they be addressed, given the scheduling constraints mentioned? What basic advice on setting up training sessions did your team ignore?...
-
Refer to Exercise 3. The mean amount of liquid in the bottles is 179.6 ml and the standard deviation is 1.3 ml. A significance test yields a P-value of 0.0589. Interpret the P-value. Exercise 3. One...
-
Consider the automobile gasoline mileage data in Table B.3. a. Build a linear regression model relating gasoline mileage \(y\) to engine displacement \(x_{1}\) and the type of transmission...
-
A project manager discovers that his team has neglected to complete the network diagram for the project. The network diagram is shown in Figure P1227. However, the project manager has some...
-
Buck Company purchased a computer and a desk for $ 9 , 0 0 0 cash. An appraiser determined the fair market value of computer to be $ 3 , 0 0 0 and the desk to be $ 7 , 0 0 0 . Based on this...
-
An aluminum master alloy manufacturer produces grain refiners in ingot form. The company produces the product in four furnaces. Each furnace is known to have its own unique operating characteristics,...
-
A toy rocket is launched straight up so that its height s, in meters, at time t, in seconds, is given by (2 points each, 4 points total) s(t) = -75t + 400t and s'(t) = -150t + 400 a) Find s'(2)
-
Consider the following two-step process: Step A has a processing time of 1 minute per unit, but no setup is required. Step B has a processing time of 0.1 minute per unit, but a setup time of 9...
-
Consider the following batch-flow process consisting of three process steps performed by three machines: Work is processed in batches at each step. Before a batch is processed at step 1, the machine...
-
Using Exhibit 12-9, determine the mix of benefits that would best fit your needs. After choosing your benefits, form into teams of three to five members and answer the following questions. Exhibit...
-
Think about the future trends in Illustration 1.6. What sorts of things will organizations have to do to respond to them? Can you think of any sectors that will not be influenced in some way by these...
-
(a) Use the Laplace transform to find the current i(t) in a single-loop LR-series circuit when i(0) = 0, L = 1 h, R = 10 , and E(t) is as given in Figure 7.3.21. (b) Use a computer graphing program...
-
Collect monthly prices for 7 (seven) stocks/assets of your choice for the last 10 (ten) years traded in a market (of your choice). Use the data to construct one portfolio (i.e. same stocks/assets)....
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
In which direction will the position of the equilibrium 2HI(g) H2(g) + I2(g) be shifted for each of the following changes? a. H2(g) is added. b. I2(g) is removed. c. HI(g) is removed. d. In a rigid...
-
a. A flask containing gaseous N2 is irradiated with 25-nm light. Using the following information, indicate what species can form in this flask during irradiation. N2(g) 2N(g) H = 941 kJ/ mol N2(g) ...
-
BAL is a chelating agent used in treating heavy metal poisoning. It acts as a bidenrate ligand. What types of linkage isomers are possible when BAL coordinates to a metal ion? CHSH CH-SH H2-OH BAL
-
Verify that the log-likelihood of model (7.7) is \(\sum_{i=1}^{k}\left[n_{i} \lambda-\exp (\lambda) ight]\). (a) Compute MLE of \(\lambda\). (b) Compute the Pearson chi-square statistic and compare...
-
Think about the general concept of a relationship, not necessarily in a business setting, but just relationships in general between any two parties. What aspects of relationships are inherently...
-
What is value? In what ways does a relationship selling approach add value to your customers, to you the salesperson, and to your sales organization?

Study smarter with the SolutionInn App



