The Haber Reaction produces ammonia NH3(g) from gaseous nitrogen and hydrogen: N(g) + 3H(g) 2NH3(g). It...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
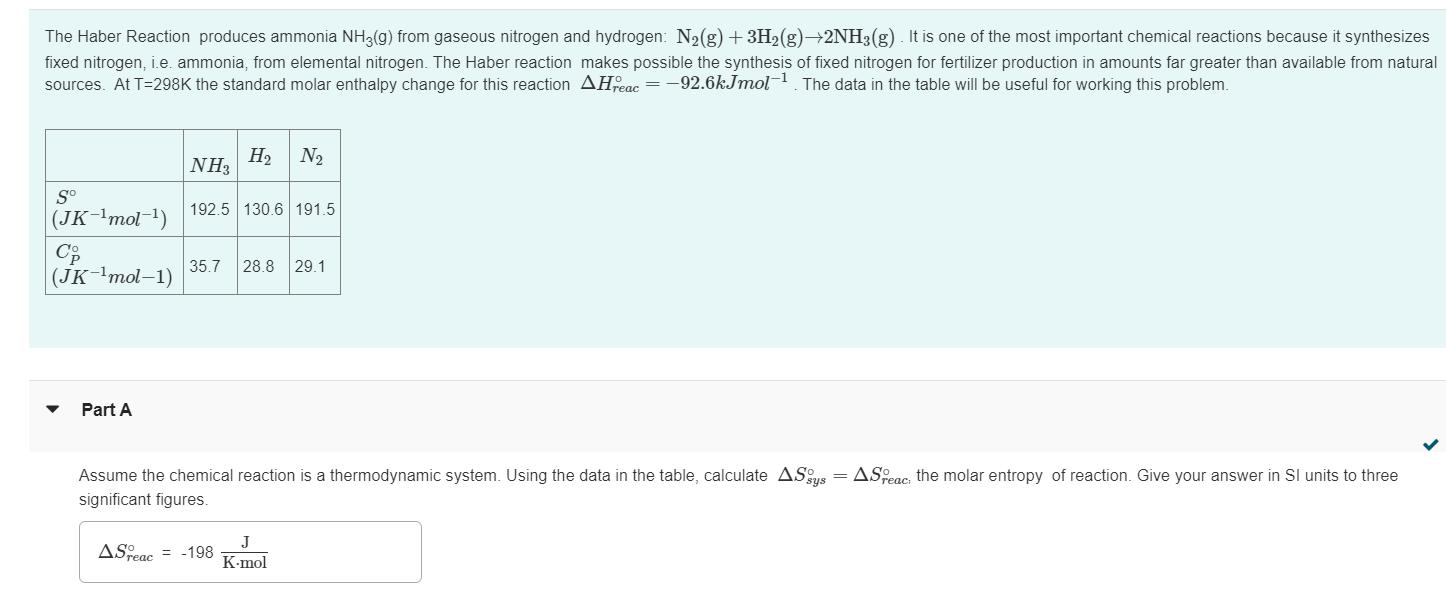
The Haber Reaction produces ammonia NH3(g) from gaseous nitrogen and hydrogen: N₂(g) + 3H₂(g) →2NH3(g). It is one of the most important chemical reactions because it synthesizes fixed nitrogen, i.e. ammonia, from elemental nitrogen. The Haber reaction makes possible the synthesis of fixed nitrogen for fertilizer production in amounts far greater than available from natural sources. At T=298K the standard molar enthalpy change for this reaction AHFeac=-92.6kJmol-¹. The data in the table will be useful for working this problem. So (JK-¹mol-¹) Co (JK-¹mol-1) Part A NH₂ H₂ N₂ 192.5 130.6 191.5 35.7 28.8 29.1 Assume the chemical reaction is a thermodynamic system. Using the data in the table, calculate ASys = ASPeac: the molar entropy of reaction. Give your answer in SI units to three significant figures. ASreac = -198 J K-mol Can the Haber reaction be made more thermodynamically favorable by increasing the temperature? To explore this question use the data in the table to calculate ASreac, the molar entropy change of the reaction at T-500K. Assume all heat capacities are constant between T-298K and T-500K. ASTeac = -221 Submit J K-mol Previous Answers The Haber Reaction produces ammonia NH3(g) from gaseous nitrogen and hydrogen: N₂(g) + 3H₂(g) →2NH3(g). It is one of the most important chemical reactions because it synthesizes fixed nitrogen, i.e. ammonia, from elemental nitrogen. The Haber reaction makes possible the synthesis of fixed nitrogen for fertilizer production in amounts far greater than available from natural sources. At T=298K the standard molar enthalpy change for this reaction AHFeac=-92.6kJmol-¹. The data in the table will be useful for working this problem. So (JK-¹mol-¹) Co (JK-¹mol-1) Part A NH₂ H₂ N₂ 192.5 130.6 191.5 35.7 28.8 29.1 Assume the chemical reaction is a thermodynamic system. Using the data in the table, calculate ASys = ASPeac: the molar entropy of reaction. Give your answer in SI units to three significant figures. ASreac = -198 J K-mol Can the Haber reaction be made more thermodynamically favorable by increasing the temperature? To explore this question use the data in the table to calculate ASreac, the molar entropy change of the reaction at T-500K. Assume all heat capacities are constant between T-298K and T-500K. ASTeac = -221 Submit J K-mol Previous Answers
Expert Answer:
Answer rating: 100% (QA)
N 8 3 4 8 2NH3 8 pant E part F T 500k AH reac 2 926... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
You are creating a customer database for the Lehigh Valley IronPigs minor league baseball team. Draw a project network given the information below. Complete the forward and backward pass, compute...
-
Most Companys close their books at the end of every calendar month; publicly-listed firms close their books not only every month but every quarter due to the SEC Requirement of the filing of a Form...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
What is the difference between a holder and a holder in due course?
-
1. Was the decision not to install the rubber bladder appropriate? Use the 5-question framework to support your analysis. 2. What faults can you identify in Fords costbenefit analysis? 3. Should Ford...
-
What are the most powerful social media networks that researchers could obtain data from about customer feedback and loyalty and why?
-
Susan Varhard, treasurer of the Gammamax Company, has proposed that the company should sell equity and buy back debt in order to maximize its value. As evidence, she presents the financial statements...
-
Cybersecurity and Infrastructure Security Agency Act Discussion 3 - Cybersecurity and Infrastructure Security Agency Act Cybersecurity and Infrastructure Security Agency Act Read through the article...
-
Street, Rhode and Close carried on business in partnership sharing profits and losses, in the ratio 5 : 4 : 3. Their draft statement of financial position as on 31 March 20X2 was as follows: Street...
-
21. Which food group was reduced the most when comparing the 1990s food pyramid to the current U.S. government's recommendations with MyPlate? Fats & Sw USE SPARINGLY MR Cheese Grup 2-3 GERVINGS...
-
Carla Esposito has an overdraft protection line. Assume that her October 2010 statement showed a latest (new) balance of $862. If the line had a minimum monthly payment requirement of 5% of the...
-
The seven basic tools for monitoring a business include all of the following except a. Pareto analysis. b. Monte Carlo statistical simulations. c. scatter plots. d. cause-and-effect diagrams.
-
Is there a difference between effective management and effective leadership? Explain.
-
Maslows hierarchy of needs a. is a theory of why people are motivated. b. uses three factors to compute the motivation of a person in a situation. c. was developed to address the Hawthorne effect. d....
-
Why are these issues sometimes difficult to talk about? What could make conversations both more comfortable and productive?
-
Using the topographic map below, answer the following questions. 1. What is the contour interval? 2. What general direction do the rivers run? 3. Imagine a profile from A to A'. Assume the profile...
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
The quantity of sulfuric acid used globally places it among the most plentiful of all commodity chemicals. In the modern chemical industry, synthesis of most sulfuric acid utilizes elemental sulfur...
-
The feed to a distillation column is a 45.0 mole% n-pentane.-55.0 mole% n-hexane liquid mixture. The vapor stream leaving the top of the column, which contains 98.0 mole% pentane and the balance...
-
The concentration of oxygen in a 5000-liter tank containing air at 1 atm is to be reduced by pressure purging prior to charging a fuel into the tank, the tank is charged with nitrogen up to a high...
-
Why is it important to talk about selling solutions instead of products or services? How does selling solutions further the success of a relationship-selling approach?
-
Has transactional selling gone the way of the dinosaur? That is, are there ever any situations in which a transactional approach to selling would be an appropriate approach today? If so, what are...
-
When a firm shifts from transactional selling to a value-added approach, a number of changes have to take place in the way a salesperson approaches customers as well as his or her own job. List as...

Study smarter with the SolutionInn App


