For a temperature of 300C and a lifetime of three years, what is the maximum stress that
Question:
For a temperature of 300°C and a lifetime of three years, what is the maximum stress that the nickel-based component of Figure 7-31 can withstand?
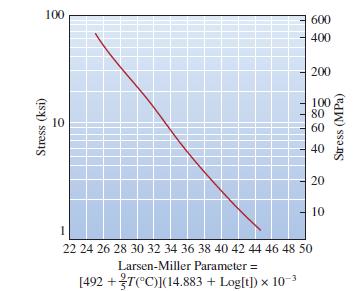
Transcribed Image Text:
100 Stress (ksi) 10 600 400 200 100 80 60 40 20 10 1 22 24 26 28 30 32 34 36 38 40 42 44 46 48 50 Larsen-Miller Parameter = [492 +T(°C)](14.883 + Log[t]) x 10-³ Stress (MPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The maximum stress 299 MPa The m...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
For a temperature of 300 K, use the equation in Problem 49 to find the bias voltage Vb for which the exponential term has the value (a) 10 and (b) 0.1.
-
What is the maximum taxable wage?
-
What is the maximum electric energy density possible in dry air without dielectric breakdown occurring?
-
which you describe and reflect on one of your favorite pop culture artifacts. Your goals will be simple: you will describe in detail a popular film, tv show, ad campaign, performer, song,...
-
Explain how a personal guarantee reduces the limited liability of the principals of a closely held corporation.
-
A steam generator evaporates \(18000 \mathrm{~kg} / \mathrm{h}\) of steam at 12 bar pressure and steam is \(97 \%\) dry. Feed water temperature \(=105^{\circ} \mathrm{C}\). Coal is fired at the rate...
-
Use the expression on page 367, involving deviations from the mean, to calculate \(r\) for the following data: x Y 3 8 6 367 -205
-
A Gallup Daily Tracking Survey found that the mean daily discretionary spending by Americans earning over $90,000 per year was $136 per day (USA today, July 30, 2012). The discretionary spending...
-
1. Over the past thirty years, post-secondary tuition rates in Canada have increased dramatically. Had tuition grown at the rate of inflation, the average in 2019-20 would have been only $2,541. What...
-
A person stands on the ball of one foot. The normal force due to the ground pushing up on the ball of the foot has magnitude 750 N. Ignore the weight of the foot itself. The other significant forces...
-
A 1-in-diameter bar of an iron-chromiumnickel alloy is subjected to a load of 2500 lb. How many days will the bar survive without rupturing at 980C?
-
A nickel-based alloy component (INCONEL alloy 601) is designed to withstand a service stress of 4000 psi at a temperature of 925C. Determine the temperature at which the component should be operated...
-
a. Find the remainder when x 3 + 6x 2 + 5x 12 is divided by i. x 2, ii. x + 3. b. Hence, or otherwise, find all the solutions to the equation x 3 + 6x 2 + 5x 12 = 0.
-
This discussion is intended to get feedback for us in designing future curricula and programs that will best suit the preparation of you and those who are coming in after you to enter a dynamic...
-
Read the Articles:"Bond market points to Fed standing firm in battle against inflation Actions" byFinancial Times (Click on thelink...
-
Create a discussion on a topic Effieciency and equity of economics based on the example given below Weekly Expectations Each week students are expected to add a discussion post in the appropriate...
-
Situation: The student has just assumed the role of manager/owner or some other type of administrative position in the area/industry for which they are majoring in. For example, a nursing student...
-
1. Do you think firms that are good at OB tend to be more profitable? Why or why not? Which of the individual mechanisms (job satisfaction; stress; motivation; trust, justice, and ethics; learning...
-
What are some uses of standard costs?
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
Hydrogen reacts explosively with oxygen. However, a mixture of H 2 and O 2 can exist indefinitely at room temperature. Explain why H 2 and O 2 do not react under these conditions.
-
The rate of the reaction depends only on the concentration of nitrogen dioxide below 225 C. At a temperature below 225 C, the following data were collected: Determine the rate law, the integrated...
-
The type of rate law for a reaction, either the differential rate law or the integrated rate law, is usually determined by which data is easiest to collect. Explain.
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...
-
Harley-Davidson, inc. (HOG), is a leading motorcycle manufacturer in the United States. The company manufactures and sells a number of different types of motorcycles, a complete line of motorcycle...
-
What is the net income for this period and total assets on this balance sheet Use the following adjusted trial balance to answer questions 22-25. Cash $ 6,530 Accounts Receivable 2,450 Prepaid...

Study smarter with the SolutionInn App


