Using the data in Figure 7-26 for an ironchromium- nickel alloy, determine the activation energy Qr and
Question:
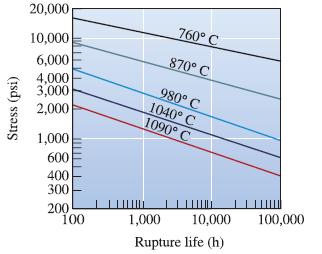
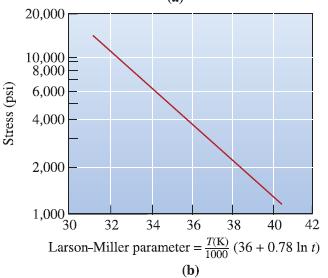
Using the data in Figure 7-26 for an ironchromium- nickel alloy, determine the activation energy Qr and the constant m for rupture in the temperature range 980 to 1090°C.
Transcribed Image Text:
Stress (psi) 20,000 10,000 6,000 4,000 3,000 2,000 1,000 600 400 300 200 100 760° C 870° C 980° C 1040° C 1090° C 1,000 10,000 Rupture life (h) 100,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The activation energy Qr HcT where Hc RT ...View the full answer

Answered By

Willis Omondi
Hi, I'm Willis Omondi, a proficient and professional academic writer. I have been providing high-quality content that best suits my clients and completing their work within the deadline. All my work has been 100% plagiarism-free, according to research from my services, especially in arts subjects and many others
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
7. Point P is located 1.7 m from fixed point charge q. The electric field E at point Pis 8.6 x 10-2 (1 point) N/C, directed away from the point charge. What is the magnitude of q? Let the...
-
Using the data in Solved Problem 10.1, prepare an activity chart like the one in the solved problem, but a second gas man also delivers 44 litres. In solved problem 10.1 Process Chart Summary O...
-
Using the data in BE21-4, compute equivalent units of production for materials and conversion costs, assuming materials are entered at the beginning of the process.
-
The management of a New York area investment firm wants to find out about the investment needs of its existing customers, for which it has an extensively detailed list, as a function of their...
-
Distinguish among a derivative action, dissent and oppression. Explain when it would be appropriate to use each of them.
-
What is meant by functional obsolescence? Could a new building suffer from functional obsolescence?
-
Express the Henry's-law constants reported in Table 8.1 as \(H_{i, p c}\) and \(H_{i, c p}\). Dimensionless temperature, 0.9 0.8 0.7 0.6 K=1+ (0-1/2) 0.5 0.4 0.3 0.2 0.1 K = 1 0 0.2 0.4 0.6 0.8...
-
Consider the following transactions for Jo Jo Music. 2013 Dec. 6 Received a $ 7,000, 90-day, 12% note in settlement of an overdue accounts receivable from Dark Star Music. 31 Made an adjusting entry...
-
Use the exact interest method (365 days) and the ordinary interest method (360 days) to compare the amount (in $) of interest for the loan. (Round your answers to two decimal places.) Principal Rate...
-
The following trial balance of Sergei Asbrecht Co. does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the...
-
Approximate the temperature at which creep deformation becomes an important consideration for each of the following metals: tin, molybdenum, iron, gold, zinc and chromium.
-
What is the difference between failure of a material by creep and that by stress rupture?
-
A clay blob of mass m 1 , initially at rest, is pushed across a frictionless surface with constant force F for a distance d. It then hits and sticks to a second clay blob of mass m 2 that is at rest....
-
Health care is a big industry in the United States, and with the baby boomers born in the late 1940s and 1950s (after World War II) starting to retire, there will be huge demands for senior health...
-
On January 15, 2020, the U.S. Treasury issues a ten-year inflation-indexed note with a coupon of 5%. On the date of issue, the CPI is 414. By January 15, 2030, the CPI index increases to 282. What...
-
Hawar International is a shipping firm with a current share price of $6 and 15 million shares outstanding. Suppose Hawar announces plans to lower its corporate taxes by borrowing $10 million and...
-
On January 20, Metropolitan, Inc., sold 8 million shares of stock in an SEO. The current market price of Metropolitan at the time was $42.50 per share. Of the 8 million shares sold, 5 million shares...
-
Dominion Virginia Power is one of the nations ten largest investor-owned electric utilities. The company delivers power to more than two million homes and businesses in Virginia and North Carolina....
-
Determine the cutoff frequency of the lowpass filter described by H() = 4/2 + j10 Find the gain in dB and phase of H() at = 2 rad/s.
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Specifications for lactated Ringers solution, which is used for intravenous (IV) injections, are as follows to reach 100. mL of solution: 285315 mg Na + 14.117.3 mg K + 4.96.0 mg Ca 2+ 368408 mg Cl -...
-
You have a solution of two volatile liquids, A and B (assume ideal behavior). Pure liquid A has a vapor pressure of 350.0 torr and pure liquid B has a vapor pressure of 100.0 torr at the temperature...
-
A solid mixture contains MgCl 2 and NaCl. When 0.5000 g of this solid is dissolved in enough water to form 1.000 L of solution, the osmotic pressure at 25.0 C is observed to be 0.3950 atm. What is...
-
The Company has 4,500,000 common shares outstanding. Jerry Fry acquired 5% of these shares at a cost of $19 each. During the current year, the Company declares a 6% stock dividend which it designates...
-
Overview of the San Antonio Federal Credit Union's current offerings, practices, and other business-related information. In this component, you will need to also provide a brief comparison between...
-
The owner of a shoe firm could instead work as an accountant to earn $300 per week. What is the implicit cost of her contribution to production?

Study smarter with the SolutionInn App


