Consider a cloud of droplets and assume that they all have the same diameter r. According to
Question:
Consider a cloud of droplets and assume that they all have the same diameter r. According to the Laplace formula (exercise 4.8), the pressure p (r) inside the droplets of radius r is related to the vapour pressure p0 (r) by,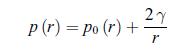
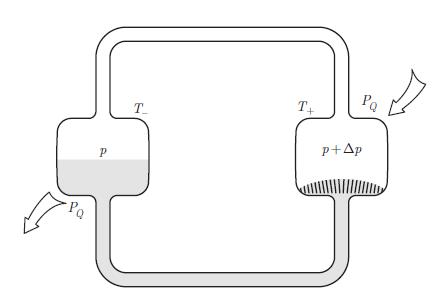
Principle of a heat pipe: At the hot side, the liquid passes through a wick and vaporises at pressure p+Δp. At the cold side, the vapour condenses at pressure p. where γ is the surface tension.We note p∞ the vapour pressure for an infinite radius. At temperature T, show that,![]()
where v is the molar volume of liquid in the limit where p∞ v RT since the molar volume of liquid is much smaller than the molar volume of gas.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:





