Conceptual Physics 12th edition Paul G. Hewitt - Solutions
Discover the comprehensive guide to mastering "Conceptual Physics 12th Edition" by Paul G. Hewitt. Unlock the potential of your studies with our online answers key, providing detailed solutions and a solution manual for every chapter. Access solved problems and step-by-step answers that cater to both students and instructors. Enhance your learning experience with our questions and answers, test bank, and instructor manual resources. Our textbook solutions pdf offers free download options, ensuring you have all the chapter solutions at your fingertips. Dive into the world of physics with confidence and clarity.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


.png)
.png)
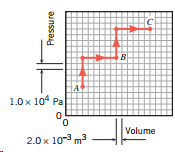
.png)
.png)
.png)
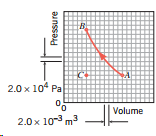
.png)
.png)
.png)
.png)