Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73. Problem
Question:
Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73.
Problem 73
Use data from Appendix IIB to calculate the equilibrium constants at 25 °C for each reaction.![]()
Appendix IIB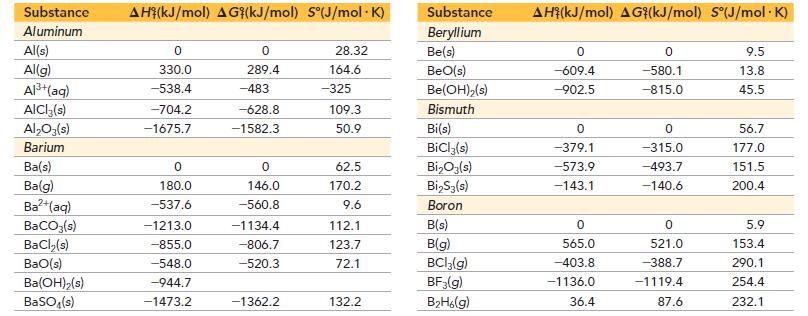
Transcribed Image Text:
a. 2 CO(g) + O₂(g) = 2 CO₂(g) b. 2 H₂S(g) = 2 H₂(g) + S₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a 190...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Estimate the value of the equilibrium constant at 655 K for each reaction in Problem 74. (H f for BrCl is 14.6 kJ/mol.) Problem 74 Use data from Appendix IIB to calculate the equilibrium constants at...
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. G f for BrCl(g) is -1.0 kJ/mol. Appendix IIB a. 2 NO(g) NO4(8) b. Br(g) + Cl(g) = 2 BrCI(g)
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix IIB a. 2 CO(g) + O(g) = 2 CO(g) b. 2 HS(g) = 2 H(g) + S(g)
-
What future directions may be interesting for comparative studies in corporate governance?
-
Explain how an activity-based budget is prepared.
-
1. Do the clinical trials in Albania. Youll be able to bring the drug to market faster and cheaper, which will be good for AH Biotechs employees and investors and good for the millions of people who...
-
Do the following activities to complete your marketing plan: 1. Draw a simple organizational chart for your organization. 2. Develop a Gantt chart (see Chapter 2) to schedule the key activities...
-
The following events took place for Air Temp Manufacturing Company during January, the first month of its operations as a producer of digital thermometers: a. Purchased $68,000 of materials. b. Used...
-
The Relational Model and Logical Design (a) In the context of the relational data model, describe the following terms: (i) relation (ii) primary key (iii) foreign key (iv) null ...
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P ICl = 2.55 atm; P I2 = 0.325 atm; P Cl2 = 0.221...
-
Describe the solubility of CaF 2 in each solution compared to its solubility in water. a. In a 0.10 M NaCl solution b. In a 0.10 M NaF solution c. In a 0.10 M HCl solution
-
What are magic numbers and why should they be avoided? How do we avoid them?
-
5)You are a consultant to a mid-sized manufacturing corporation that is considering an investment project. The project requires an initial investment of $100 million and will generate an after tax...
-
Gleam Company has a line of successful dental products that are sold in retail outlets across the country. These products include toothpastes, flosses, and rinses. The financial managers of Gleam are...
-
George and Claire are married and have 4 children. If they elect gift splitting, what is the maximum amount of total gifts they can make to their children (without using a 529 plan) in 2023 and have...
-
Mineral oil with given sp.gr 0.96 flows-up through an inclined pipe of uniform diameter 220mm with a mean velocity of 1.2 m/s. The pressure at the datum of the pipe is measured as 30 N/cm2 with a...
-
Analyze the factors influencing enterprise value and elucidate their impact on organizational valuation.
-
What is a common body of knowledge?
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
Redraw the ray diagram for a compound microscope (Fig. 5.110), but this time treat the intermediate image as if it were a real object. This approach should be a bit simpler. Fig. 5.110 Exit pupil fe...
-
Consider a thin positive lens L 1 , and using a ray diagram, show that if a second lens L 2 is placed at the focal point of L 1 , the magnification does not change. Thats a good reason to wear...
-
Draw a ray diagram locating the images of a point source as formed by a pair of mirrors at 90? (Fig. P.5.61a). Now create a ray diagram locating the images of the arrow shown in Fig. P.5.61b. Fig...
-
Perform the indicated operations. Express your answer in reduced form. 9 2 (x-5)(x + 1) x-5
-
rephrase: Secondly, financial resources are crucial to cover expenses such as instructional materials, technology tools, and any additional costs associated with implementing the module. Careful...
-
What events of the 1980s prompted public demand for governments to "get tough" on fraudulent businesses and negligent accountants? a. stock market crash b. bank failures in the U.S. c. layoffs of...

Study smarter with the SolutionInn App


