Use the data in Figure 13.1 to fi nd the instantaneous rate of disappearance of the reactant
Question:
Use the data in Figure 13.1 to fi nd the instantaneous rate of disappearance of the reactant at 20 seconds.
Figure 13.1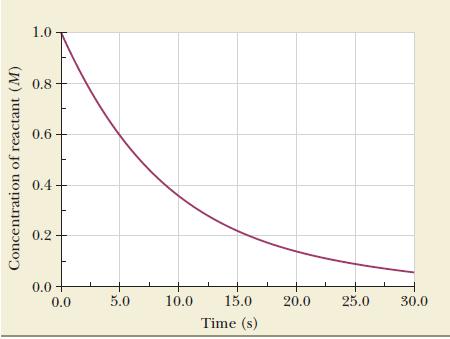
Transcribed Image Text:
Concentration of reactant (M) 1.0 0.8 0.6 0.4 0.2 0.0 + 0.0 5.0 10.0 15.0 Time (s) 20.0 25.0 30.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Ra...View the full answer

Answered By

Hillary shikhuma
I have 21 years experience preparing students for Cambridge IGCSE and A level Chemistry and Biology. I have more interest in teaching chemistry however, my students always testify they enjoy my biology classes more. I use exam-focused resources to present key concepts and applications and use exam style questions to practice with the students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Figure 13.1 to calculate the instantaneous rate of disappearance of the reactant at 10 seconds. Strategy The instantaneous rate is the slope of the tangent, so place a ruler on the...
-
The data in Figure 7.15, Table 7.19, and Table 7.20 are provided for end items A and H. Develop MRP records for items B, C, I, and E. Are there any action notices for the first week? In figure 7.15 ...
-
In the following exercises, you will use the data in the TAL Distributors database shown in Figure 2-1 in Chapter 2. (If you use a computer to complete these exercises, use a copy of the original TAL...
-
Ronnie owns 600 shares of a stock mutual fund. This year he received dividend distributions of 60 stock mutual fund shares ($ 40 per share) and long term capital gain distributions of 45 stock mutual...
-
The Hamiltonian operator for a two-state system is given by, H = (| l> <1 | |2 > < 2| + |1 > < 2| + |2 > < 1|), where a is a number with the dimension of energy. Find the energy Eigen values and...
-
Caren Smith opened a medical practice. During July, the first month of operation, the business, titled Caren Smith, M. D., experienced the following events: July 6 Smith contributed $ 55,000 in the...
-
Does life satisfaction change by region over time? Use the lifesat_childmort data from Example 2.15 to explore this question. Data from Example 2.15 The World Happiness Report [Helliwell et al.,...
-
Suppose you are the manager of a fitness center that is one of many in a chain. Give one example of a cost that you control and one example of a cost you do not control. Why is it important in this...
-
Find g'(x) where, 9(x)=3x3 5 5
-
When 0.030 mol HCl dissolves in 100.0 g benzene, the solution freezes at 4.04 C. When 0.030 mol HCl dissolves in 100.0 g water, the solution freezes at -1.07 C. Use the data in Table 12.4 to complete...
-
From the heating curves, which of the two substances, A or B, has the greater enthalpy of fusion?
-
What is the general trend in electronegativity within a group of elements in the periodic table? Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca...
-
ASC 270 standardized the preparation and reporting of interim income statements by Multiple select question. measuring the contingent liabilities as per the footnotes in the interim report. defining...
-
What document will an administrator of an estate generally need to access assets that belonged to a decedent? Briefly explain.
-
Explain and justify how you would calculate planning materiality for lululemon. Calculate an appropriate $ amount for the planning materiality. I identified some accounts that may be at risk of...
-
Explain the budgeted income statement. Outline how you would create annual budgeted financial statements. Analyze the difference between a static budget and a flexible budget, including the...
-
Explain the main items commented on and explain their importance. Real-life example: Refer to financial commentary on a specific company (Nike) to highlight how analysts emphasize certain financial...
-
Panner, Inc owns 30% of Watkins & applies the equity method. During the current year Panner buys inventory costing 54,000.000 and sells it to Watkins for 90,000.0. At the end of the year Watkins...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Calculate the mass of a body in kg if it has a kinetic energy of 38.6 Nm when moving at 31.5 km/h. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the mass of a body in g if it has a kinetic energy of 94.6 m Nm when moving at 2.25 m/s. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the velocity in m/s of a 12-kg object if it has a kinetic energy of 15 Nm. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Some might argue whether humanity is evil or good. First, one must ask what is good. What is evil? You save your child from death and despair by killing another person with a will to kill them...and...
-
ABC Company receives a document from Teresa Smith which states, "Forty-five days after the undersigned date, I promise to pay $2,000.00 (two thousand dollars and no cents) at a rate of 8.75% interest...
-
What is business law? How it can be implemented perfectly.

Study smarter with the SolutionInn App


