209 Bi is the heaviest stable nuclide, and its BE/A is low compared with medium-mass nuclides. Calculate
Question:
209Bi is the heaviest stable nuclide, and its BE/A is low compared with medium-mass nuclides. Calculate BE/A, the binding energy per nucleon, for 209Bi and compare it with the approximate value obtained from the graph in Figure 31.24.
Data from figure 31.24
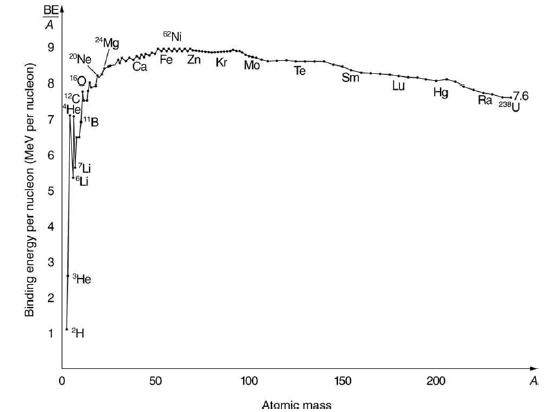
Transcribed Image Text:
Binding energy per nucleon (MeV per nucleon) BE A 9 8 6 5 4 3 2 20 Ne 160 0 He 1 ¹2H 24Mg Ca 50 62 Ni Zn Kr Mo 100 Te Atomic mass Sm 150 LU Hg 200 7.6 Ra 2U A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
7848 M...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
(a) Show that the total binding energy Ebe of a given nuclide is Ebe = ZH + Nn - , where H is the mass excess of 1H, n, is the mass excess of a neutron, and A is the mass excess of the given nuclide....
-
2H is a loosely bound isotope of hydrogen. Called deuterium or heavy hydrogen, it is stable but relatively rare-it is 0.015% of natural hydrogen. Note that deuterium has Z = N, which should tend to...
-
The binding energy per nucleon for most nuclides doesnt vary much (see Fig. 43.2). Is there similar consistency in the atomic energy of atoms, on an energy per electron basis? If so, why? If not, why...
-
Figure shows an overhead view of a ring that can rotate about its center like a merry-go-round. Its outer radius R2 is 0.800 m, its inner radius R1 is R2/2.00, its mass M is 8.00 kg, and the mass of...
-
LeeJin Corp. started a two-year period with 200,000 shares of $2 par common stock outstanding. During the two years, LeeJin had the following capital stock transactions: (a) Declared a 10% common...
-
Television and radio waves reflecting from mountains or airplanes can interfere with the direct signal from the station. (a) What kind of interference will occur when 75-MHz television signals arrive...
-
On February 1, Piscina Corporation completed a combination with Swimwear Company accounted for as a pooling of interests. At that date, Swimwears account balances were as follows: Inventory. Land....
-
Weinstein, McDermott, and Roediger (2010) report that students who were given questions to be answered while studying new material had better scores when tested on the material compared to students...
-
Sales-related transactions, including the use of credit carde Instruction Chart of Account ma ruction Are we C Swan VMA 15.000 200 De wet. The two E o ator Co. Ma VISA MER Sales-related transactions,...
-
The purpose of this problem is to show in three ways that the binding energy of the electron in a hydrogen atom is negligible compared with the masses of the proton and electron. (a) Calculate the...
-
Unusual patterns always indicate the existence of fraud. (True/False)
-
What were good and what were wrong in the U.S. government's responses to the crisis? (Subprime Mortgage Crisis)
-
10.) Steam enters a well-insulated turbine at 6 MPa, 400C and expands to 200 kPa, saturated vapor at a rate of 10 kg/s. (a) Draw a schematic of the process (5 pts). (b) Determine the exergy...
-
4. [8 marks] The tides in the Bay of Fundy are some of the largest in the world. The height, h(t), of the tide in meters after t hourse can be modeled by 39 h(t) = 25 con (77) + 30 4 COS 6 (a) What...
-
Wolfe, Inc. had credit sales for the period of $144,000. The balance in Allowance for Doubtful Accounts is a debit of $653. If Wolfe estimates that 2% of credit sales will be uncollectible, what is...
-
Water at 20C is to be pumped from a reservoir (ZA = 5 m) to another reservoir at a higher elevation (ZB = 13 m) through two 36-m- long pipes connected in parallel as shown. The pipes are made of...
-
Delph Company uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company estimated that 53,000 machine-hours would...
-
Write the augmented matrix for each of the following systems of linear equations. (a) x - ly = 5 2x + y = 1 (b) x + 2y = 0 y = 1 (c) x - y + z = 2 x - z = l y + 2x = 0 (d) x + y = 1 y + z = 0 z - x -...
-
7 A 29-year-old, previously healthy man suddenly collapses at a party where legal and illicit drugs are being used. Enroute to the hospital, he requires resuscitation with defibrillation to establish...
-
Make a sketch of how the position and kinetic energy of a harmonic oscillator vary with time. The period of the oscillations of KE can be determined from the separation in time of adjacent maxima in...
-
In Section 11.3, we discussed the total mechanical energy of a mass-on-a-spring oscillator. The result in Equation 11.21 shows that the total energy is proportional to the square of the amplitude....
-
Use energy considerations to derive the oscillation frequency for a mass-on-a-spring oscillator. The maximum potential energy stored in the spring must be equal to the maximum kinetic energy of the...
-
A company manufactures lawnmowers. Compute the total amount of period costs from thr following costs.
-
TestAnswerSavedHelp opens in a new windowSave & ExitSubmit Item 1 7 1 0 points Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1 : 2 0 : 1 8 Item 1 7 Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1...
-
Use the following information for the Problems below. (Algo) [The following information applies to the questions displayed below.] Lansing Company's current-year income statement and selected balance...

Study smarter with the SolutionInn App


