Find the following for path A in Figure 2.57: (a) The distance traveled. (b) The magnitude of
Question:
Find the following for path A in Figure 2.57:
(a) The distance traveled.
(b) The magnitude of the displacement from start to finish.
(c) The displacement from start to finish.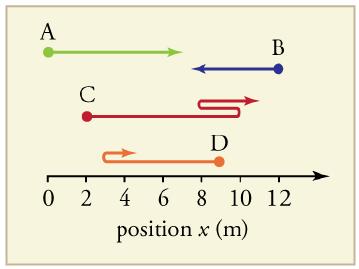
Transcribed Image Text:
A C D B 02 4 6 8 10 12 position x (m)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a ...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Alyeski Tours operates day tours of coastal glaciers in Alaska on its tour boat the Blue Glacier. Management has identified two cost drivers-the number of cruises and the number of passengers- that...
-
You have fit a k-means algorithm with six clusters. The results are visualized below. Based on the plot, which statement is true? There is overlap between some clusters, which indicates there is no...
-
Find the following for path A in Figure 3.54: (a) the total distance traveled, and (b) the magnitude and direction of the displacement from start to finish.
-
Modify BST to add a method rangeSearch () that takes two keys as arguments and returns an iterable over all keys that are between the two given keys. The running time should be proportional to the...
-
The following data were taken from the financial statements of Orion Systems Inc. for the current fiscal year: Assuming that total assets were $2,009,000 at the beginning of the year, determine the...
-
Figure P5.46 shows a lateral pipe fitting. This particular fitting has a mainline diameter of \(4.0 \mathrm{in}\). The diameter of the lateral is \(3.0 \mathrm{in}\)., and the lateral angle is...
-
A liquid in equilibrium with its own vapour at a specified temperature or pressure is called a (a) Superheated liquid (b) Saturated liquid (c) Sub-cooled liquid (d) None of these.
-
Abs Health Club has the following cost equation: Total Costs = $40,000 + $80n, Where n = number of memberships. a. Calculate Abss average fixed cost per membership when there are 1,000 memberships....
-
What is trust, and how does it relate to justice and ethics? Do you agree that smartphone use has decreased the kinds of reflection that might deepen ethical maturity? Do you think that society has...
-
What are the magnitude and sign of displacements for the motions of the subway train shown in parts (a) and (b) of Figure 2.18? Data given in Figure 2.18 Strategy A drawing with a coordinate system...
-
Jerry Von Rohr was employed as the president, chairman, and CEO of Reliance Bancshares Inc. In September 2001, Von Rohr and Reliance entered into an amended employment agreement which included that...
-
Adams Leasing (lessor) agrees on January 1, 2019, to rent Healthy Diner (HD) (lessee) the equipment that HD requires to expand its restaurant. The lease agreement calls for 10 annual lease payments...
-
Consider a crude extract with a mixture of the 5 proteins listed below. Protein A 4 . 5 pI 1 6 0 molecular weight, kDa Protein B 1 2 . 5 pI and 6 5 molecular weight Protein C 5 . 0 pI , 1 5...
-
Changes to temperature: Endothermic reaction: Exothermic reaction: 8. reactants + heat products reactants products + heat The reaction: 2SO2(g) + O2(g) 2SO3(g) 160. AH = positive = negative...
-
Iodine is sparingly soluble in pure water. However, it does ` dissolve ' in solutions containing excess iodide ion because of the following reaction: I - ( aq ) + I 2 ( aq ) = I 3 - ( aq ) K = 7 1...
-
O Macmillan Learning The element carbon has two naturally occurring isotopes. The isotopic masses and abundances of these isotopes are shown in the table. Isotope Isotopic mass (u) 12 C 12.00 13 C...
-
Determine the oxidation states for each of the following elements or ions. Zn: +2 S-: -2 S: -2 Fe+: +2
-
In 2017, Taymouth Inc. reported net income of $100,000. What would be the effect on retained earnings on Taymouth's 2017 balance sheet and on its 2018 income statement if it didn't record a closing...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Record the following purchase transactions of Custom Kitchens Inc. Oct. 6 Oct. 9 Oct. 20 Purchased 230 cabinet doors on credit at a cost of $46 per door. Shipping charges are an extra $2 cash per...
-
Record the following purchase transactions of Money Office Supplies. Aug. 3 Aug. 7 Aug. 12 Purchased 45 chairs on credit, at a cost of $55 per chair. Shipping charges are an extra $3 cash per chair...
-
Which of the following accounts are used when recording a purchase? A. Cash, merchandise inventory B. Accounts payable, merchandise inventory C. A or B D. Cash, accounts payable
-
1. A 115 volt fluorescent lamp is rated at 0.43 amps (430 milliamperes). Find the watts that will be consumed by the lamp and its ballast. (115 volts is specified instead of 120 volts to allow for...
-
1. What shape (function) does the graph seem to follow? Does it agree with the expected theory for radioactive decay? 2. If you started with a sample of 600 radioactive nuclei, how many would remain...
-
Selected balance sheet and income statement information from a company's Form 10- K is as follows: Consolidated Balance Sheet As of December 31 2021 2020 Cash 5,820 4,170 Marketable Securities...

Study smarter with the SolutionInn App


