Under what temperature conditions would the following reactions occur spontaneously? (a) 2 NH4NO3(s) - (b) I2(g) 2
Question:
Under what temperature conditions would the following reactions occur spontaneously?
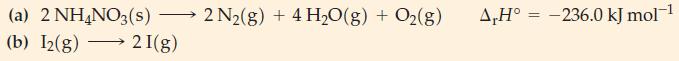
Transcribed Image Text:
(a) 2 NH4NO3(s) - (b) I2(g) 2 I(g) 2 N₂(g) + 4 H₂O(g) + O2(g) A,H° -236.0 kJ mol-¹ =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Analyze a The reaction is exothermic and in Example 132a we concluded that S 0 because large quantit...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Which of the four cases in Table 13.3 would apply to each of the following reactions? (B) Under what temperature conditions would the following reactions occur spontaneously? (a) The...
-
Which of the following reactions occur spontaneously as written, with the production of a measurable electric current? a. I 2 + NaBr Br 2 + 2NaI b. Li + NaCl LiCl + Na c. Li + + Na + NaLi d. Ag +...
-
Using data from Appendix 4, calculate ÎHo, ÎSo, and DGo for the following reactions that produce acetic acid: Which reaction would you choose as a commercial method for producing acetic...
-
As mentioned in Section 5.6, Sainte-Venants principle will allow particular boundary conditions to be replaced by their statically equivalent resultant. For problems (b), (c), (d),and (f) in Exercise...
-
Edington Electronics Inc. produces and sells two models of pocket calculators, XQ-103 and XQ-104. The calculators sell for $15 and $25, respectively. Because of the intense competition Edington...
-
The man picks up the 52-ft chain and holds it just high enough so it is completely off the ground. The chain has points of the attachment A and B that are 50 ft apart. If the chain has a weight of 3...
-
Why is an ethical culture considered necessary for fraud prevention and deterrence?
-
Lashkova Company had accounts receivable of $100,000 on January 1, 2014. The only transactions that affected accounts receivable during 2014 were net credit sales of $1,000,000, cash collections of...
-
Statement of cash flows-indirect method The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: December 31, 20Y9 December 31, 20Y8 Assets Cash Accounts...
-
Arrange the entropy changes of the following processes, all at 25 C, in the expected order of increasing S, and explain your reasoning: (a) HO(1, 1 bar) (b) CO (s, 1 bar) (c) HO(1, 1 bar) 111 HO(g, 1...
-
Indicate whether each of the following changes represents an increase or a decrease in entropy in a system, and explain your reasoning: (a) The freezing of ethanol; (b) The sublimation of dry ice;...
-
Your grandfather put some money in an account for you on the day you were born. You are now 18 years old and are allowed to withdraw the money for the first time. The account currently has $3996 in...
-
What is the problem of using survey results?
-
A trend equation is a regression equation in which a. there are multiple independent variables. b. the intercept and the slope are the same. c. the dependent variable is time. d. the independent...
-
How can quantitative analysis techniques inform and support decision making within organizations?
-
An analysis to determine how much a solution would change if there were changes in the model or the input data is called a. sensitivity or postoptimality analysis. b. schematic or iconic analysis. c....
-
It is often said that linear programming is not the most suitable tool for addressing business management problems. a. True b. False
-
Arts N Crafts Industries manufacturers high-end light fixtures which it sells internationally. The company is responsible for paying for shipping its product to a distributor located in Atlanta,...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Backflush, two trigger points, completion of production and sale (continuation of 20-33). Assume the same facts as in Problem 20-33 except now there are only two trigger points: the completion of...
-
Lean Accounting. Flexible Security Devices (FSD) has introduced a just-in-time production process and is considering the adoption of lean accounting principles to support its new production...
-
Backflushing. The following conversation occurred between Brian Richardson, plant manager at Glendale Engineering, and Charles Cheng, plant controller. Glendale manufactures automotive component...
-
1. Consider a CPU with a simple non-pipelined data path. The registers A, B, MDR and the ALU are all 8-bit wide. SP and MAR are 16-bit registers. Each memory operation takes 2 CPU clock cycles and...
-
Q1. (a) A solid block of timber strut of density 850 kg/m and volume 0.6 m floats in a liquid of density 1250 kg/m. i. What is the mass of the block?. (3 marks) ii. What is the weight of liquid...
-
Consider the following probability distribution for stocks C and D: State Probability Return on Stock C Return on Stock D 1 0.30 2 0.50 3 0.20 7% 11% -16% -9% 14% 26% Compute the correlation...

Study smarter with the SolutionInn App


