Consider the use of CF2Cl2 as a dispersing agent for aerosol spray cans. Estimate the pressure a
Question:
Consider the use of CF2Cl2 as a dispersing agent for aerosol spray cans. Estimate the pressure a can has to withhold at 40°C. Its enthalpy of vaporization at its normal boiling point (244 K) is 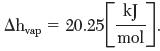
State your assumptions.
Transcribed Image Text:
Ahvap 20.25 kJ mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The Company XYZ has 1173 blocks of building for its business operation, where each block has 7 floors. The distance between each floor is 7 meters. ] (ii) Give a function run2diff which can be...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The Compiler Construction Programming answers should be written in some notation approximating SML or OCaml. (a) Describe what is meant by tail recursion. [4 marks] (b) Eliminate tail recursion from...
-
For each of the following tests, identify two different samples of people who would have the expertise to serve as subject matter experts (SMEs) for providing judgments regarding the content validity...
-
Write a paper on "Should regulations regarding the use of cell phones be standardized"
-
General Bonds Corp. (GBC) has a pension liability of $100M. The duration of this liability is 15 years. On the asset side of its balance-sheet, GBC has a 100% bond portfolio that consists of two...
-
Let the real-valued random process \(U(t)\) be defined by \[ U(t)=A \cos (2 \pi v t-\Phi) \] where \(v\) is a known constant, \(\Phi\) is a random variable uniformly distributed on \((-\pi, \pi), A\)...
-
Ybor City Tobacco Company has for many years enjoyed a moderate but stable growth in sales and earnings. In recent times, however, cigar consumption and consequently Ybors sales have been falling,...
-
You are a Morgan Stanley portfolio manager of a risky portfolio with an expected rate of return of 19% and a standard deviation of 28%. The T-bill rate is 7%. Suppose your client decides to invest in...
-
At 1 atm titanium melts at 1941 K and boils at 3560 K. Its triple point pressure is 5.3 Pa. Using only these data, estimate the enthalpy of vaporization of titanium. You will need to think about a...
-
A TP diagram of carbon is presented in the following fi gure. The following data are available at 25C. Answer the following questions: (a) Identify the region where diamond is the thermodynamically...
-
Use the contingency table and expected frequencies from Exercise 11. At = 0.10, test the hypothesis that the variables are independent. Perform the indicated chi-square independence test by...
-
How do social network analysis techniques elucidate the patterns of communication, influence, and collaboration within teams, and how can this knowledge be leveraged to enhance information flow,...
-
Your text discusses the concept of groupthink. Discuss a time when you have experienced groupthink.Discuss key characteristics of groupthink that were present in your experience. Then identify at...
-
Monica is a cash basis, self-employed chef with her own restaurant. She didnt receive any 1099-Misc forms, but her total deposits in her business bank account for 2017 show $151,723. In addition to...
-
Consider the function fix) = -x on (0,2T] and its 211-periodic extension f(x) by f(x) = f(x-2R) for XE (2KIT, 2 (KHXT] V REZZ Sketch f, find its Fourier series, and discuss the pointuise convergence....
-
Mahalo Boat Adventure Inc. has a July 31 year-end. It showed the following partial amortization schedules regarding two bond issues: Bond Issue A Period Ending June 1/20 Dec. 1/20 Dec. 1/26 June 1/27...
-
Name five characteristics of goodwill messages.
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Ozone absorbs ultraviolet radiation in a part of the electromagnetic spectrum energetic enough to disrupt DNA in biological organisms and that is absorbed by no other abundant atmospheric...
-
G.C.G. Wachewsky, R. Horansky, and V. Vaida (J. Phys. Chem. 100, 11559 (1996)) examined the UV absorption spectrum ofCH3I, a species of interest in connection with stratospheric ozone chemistry. They...
-
One of the principal methods for obtaining the electronic spectra of unstable radicals is to study the spectra of comets, which are almost entirely due to radicals. Many radical spectra have been...
-
Identify and explain conceptual connections between the film Alien (1979) and Breeding Influenza: The Political Virology of Offshore Farming by Robert G. Wallace. a. Where do the author's...
-
Regarding an affordable housing project in rhode island: Describe and itemize the equipment requested for purchase, the intended purpose of each item, and how the estimated costs were determined....
-
What is the suggested approach for increasing participation in worksite health programs?

Study smarter with the SolutionInn App


