Question: (a) Use these data to plot solubility as a function of temperature for KCl and Li 2 SO 4 : (b) Using the plot, estimate
(a) Use these data to plot solubility as a function of temperature for KCl and Li2SO4:
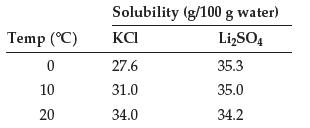
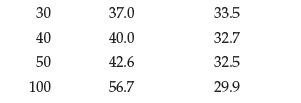
(b) Using the plot, estimate the solubility of both compounds in water at 70°C.
(c) How much of each compound can be dissolved in a beaker containing 75 g of water at 70°C?
Temp (C) 0 10 20 Solubility (g/100 g water) LigSO4 27.6 31.0 34.0 35.3 35.0 34.2
Step by Step Solution
3.39 Rating (149 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


