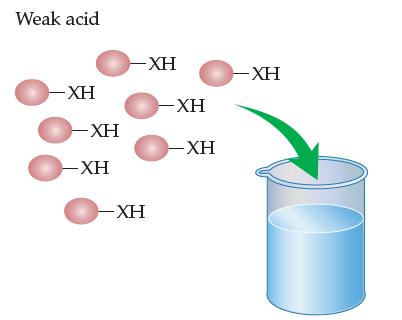
Imagine that the eight identical molecules shown below are of a weak molecular acid. Draw a picture
Question:
Imagine that the eight identical molecules shown below are of a weak molecular acid. Draw a picture showing all the expected ions and/or molecules present after the molecules dissolve in the water in the beaker and equilibrium is reached.
Transcribed Image Text:
HX- HX- HX- HX- HX- HX HX- HX- рре урам
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The beaker on the left below represents a buffer solution of a weak acid HA and its conjugate base, A. (Water molecules and spectator ions have been omitted for clarity.) a. Which beaker on the...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
A saturated solution of Cd(OH)2 is shown in the middle beaker. If hydrochloric acid solution is added, the solubility of Cd(OH)2 will increase, causing additional solid to dissolve. Which of the two...
-
The compound bow in the previous problem requires 152 J of work to draw the string back to x = 0.540 m, at which point the archer need only exert 250 N of force to hold the string in place. (a)...
-
Preparing a Bank Reconciliation and Related Journal Entries The bookkeeper at Wood Company has not reconciled the bank statement with the Cash account, saying, I dont have time. You have been asked...
-
Using the data in E prepare a statement of cost of goods manufactured for the month ended January 31, 2016, assuming that no further work was done in Immersion during January. In E, Alpine Adhesives...
-
In July 2008, Brian, Dale, and Sandra Allen signed a contract with East Resources, Inc., concerning 148 acres of the Allens property. East wanted to develop and exploit the oil and gas resources...
-
Wayne Hills Hospital in tiny Wayne, Nebraska, faces a problem common to large, urban hospitals as well as to small, remote ones like itself. That problem is deciding how much of each type of whole...
-
Daniela built a raised garden bed that is 15 feet long, 6 feet wide, and 2.5 feet tall. She plans to fill the garden bed with premium soil that costs $15.96 per cubic yard. Recall that 1 yard = 3...
-
Answer Problem 15.80 for the case where the eight molecules are of a strong acid. Strong acid HX- HX- HX- -XH -XH -XH -XH HX-
-
Other than water, which species would you expect to find in the highest concentration in an aqueous solution of CH 3 COOH? Explain.
-
Find the center and radius of the sphere with equation .r2 + y2 + z2 - 6x + 2y - 8z = 0.
-
Question 1: Art and Science College (ASC) is a private college established in 2016. The college is an educational institution approved by the Minister of Education. ASC offers a new diploma in...
-
Van Westendorp analysis is a data analytic technique relying on 4 survey questions that can be used to identify the optimal pricing for an offering A) True B) False
-
1. Global Thoughts (Discussion Board) (20 points): In two or three paragraphs (maximum), briefly address the following: . You are an entrepreneur and have your own company (choose any legitimate...
-
The following data apply to Stetson and Associates (raillions of dollars) Cash Net Fixed Assets Sales Net Income ROE Quick Ratio Current Ratio DSO Assers 4400 $4.300 1,400,000,000 TA30,000 444.600...
-
Define the concept of services marketing. Explain how services marketing differs from product marketing and describe some of the challenges that service marketers face. Then choose a specific company...
-
Your restaurant is located near a high school. Recently, several of the students who are patrons have been throwing ice and wadded paper napkins at each other. What should you do?
-
Organizations are increasing their use of personality tests to screen job applicants. What are some of the advantages and disadvantages of this approach? What can managers do to avoid some of the...
-
Compute the pressure produced in the oil in a closed cylinder by a piston exerting a force of 12.0 kN on the enclosed oil. The piston has a diameter of 75 mm.
-
A hydraulic cylinder must be able to exert a force of 38.8 kN. The piston diameter is 40 mm. Compute the required pressure in the oil.
-
The hydraulic lift for an automobile service garage has a cylinder with a diameter of 8.0 in. What pressure must the oil have to be able to lift 6000 lb?
-
Why has the healthcare industry USUALLY been a slow adopter of new technologies compared to other industries? Do you see this changing in coming years? Why or why not?
-
What would a more comprehensive CSR plan for Host Europe look like? What kind of measures, and tracking could be introduced, without putting Host Europe's economic success at risk?
-
List the ways in which your organization is involved with intellectual property. You do not have to provide any trade secrets or specifics, but speak to where intellectual property is important to...

Study smarter with the SolutionInn App


