Question: Write the equilibrium constant expression for (a) SnO(s) + 2 H(g) Sn(s) + 2 HO(1) (b) H3PO4(aq) + 3 HO(1) PO (aq) 2+ (c) Pb+
Write the equilibrium constant expression for
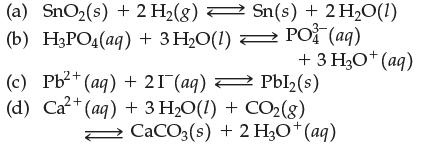
(a) SnO(s) + 2 H(g) Sn(s) + 2 HO(1) (b) H3PO4(aq) + 3 HO(1) PO (aq) 2+ (c) Pb+ (aq) + 21 (aq) +3HgO*(aq) Pbl(s) (d) Ca+ (aq) + 3 HO(1) + CO2(g) CaCO3(s) + 2 H3O+ (aq)
Step by Step Solution
3.33 Rating (156 Votes )
There are 3 Steps involved in it
The equilibrium constant expression for the reaction in the image is Kp SnO2s H2g2 Sns H2Og2 Where K... View full answer

Get step-by-step solutions from verified subject matter experts


