0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is...
Question:
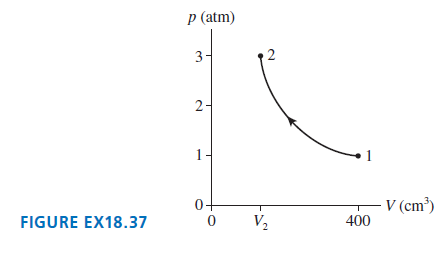 a. What type of process is this?
a. What type of process is this?
b. What is the final temperature in °C?
c. What is the final volume V2?
Transcribed Image Text:
p (atm) 3- 2- 1- V (cm³) 400 0+ V2 FIGURE EX18.37 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
Model Assume that the gas is an ideal gas Solve a The graph shows that th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.? What are (a) Temperature T 1 , (b) Pressure p 2 , (c) Volume V 3 ? p (atm) P2 - T = 2926 K 3 T3 = 2438 K 1 0+ V (cm) V3...
-
A gas undergoes the process shown in FIGURE Q18.10. By what factor does the temperature change? FIGURE Q18.10
-
Mr. Mo carries on business as a sole proprietor. The fiscal year end of the business is December 31. During 2020, its first year of operation, net business loss amounts to $72,000. In addition, the...
-
Before Lemon Corporation was taken private in a transaction engineered by its largest stockholder, some of Lemon's employees had unexercised options to purchase stock of Lemon. Under an employee...
-
In 2002, the journal Science reported that a study of women in Finland indicated that having sons shortened the life spans of mothers by about 34 weeks per son, but that daughters helped to lengthen...
-
Jan Samson, CEO at CalleetaCO, sat staring at the now-empty boardroom. Her board of directors had reacted negatively to Jans growth proposals for expanding CalleetaCO globally, leaving Jan with a big...
-
A typical coal-fired power plant generates 1000 MW of usable power at an overall thermal efficiency of 40%. (a) What is the rate of heat input to the plant? (b) The plant bums anthracite coal, which...
-
A Corporation wants to raise $1,210,000 via a rights offering. The company currently has 220,000 shares of common stock outstanding that sells for $32 per share. The issue will allow current...
-
? Dean and Ellen Price are married and have a manufacturing business. They bought a piece of business equipment (7-year personal property) on 4/1/2017 for $50,000. Use half-year convention to...
-
A gas with an initial temperature of 900°C undergoes the process shown in FIGURE EX18.36. a. What type of process is this? b. What is the final temperature in °C? c. How many moles of gas are...
-
iRobot designs and manufactures robots for consumer, commercial, and military use. For the fiscal year ended January 2, 2016, the company reported the following on its balance sheet and income...
-
Graph the following observations of \(x\) and \(y\) on graph paper. a. Using a ruler, draw a line that fits through the data. Measure the slope and intercept of the line you have drawn. b. Use...
-
In October 2012, Michael Eisenberg and Miriam Weisbecker executed a purported joint venture agreement with Gerard Rem wherein Eisenberg advanced \($220,000\) to secure a 50 percent interest in the...
-
Robert Law, in his capacity as a trustee for a trust, asked the trial court to enter a charging order under ORS 70.295 against the four limited partnerships that Ronald Zemp was partner of for Zemps...
-
Heartland State Bank routed eight checks to a collections bank, Federal Reserve. Federal Reserve then delivered the checks and additional cash letters to the defendant bank, American Bank & Trust, on...
-
When GTE Sylvania discovered it was losing market share to other television manufacturers, it developed a franchise plan that limited the number of retailers selling its product in each area....
-
In 2005, Ann Furey obtained a loan secured by a mortgage from Merchants Bank to purchase a camp in Lincoln, Vermont. In August 2009, she stopped making mortgage payments. In April 2011, the bank...
-
The following table presents a hypothetical panel of data: a. Estimate the groupwise heteroscedastic model of Section 9.7.2. Include an estimate of the asymptotic variance of the slope estimator. Use...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Make a very rough estimate of the thermal energy content of Earth, assuming that the core has radius 3480 km, temperature 4000 K, density 11000 kg/m 3 and heat capacity 800 J/K kg, and that the rest...
-
Check eq. (32.4) by estimating the radioactive heat production rate using data from Table 20.6 Take the fractional abundances of 235 U, 40 K to be 0.72%, 0.012%. Take the average energy release in 40...
-
Assume that a region of continental crust has a typical surface heat flux of 65 mW/m 2 , crustal density of 2750 kg/m 3 , and typical crustal abundances of radioactive nuclides (as given in Table...
-
If Olivia is single and in the 24 percent tax bracket, and she sold stock for $12,000 that she purchased for $10,000, 9 months earlier. What should her tax expense be in 2018?
-
what current trends at HP computer and electronics company should an investor review?
-
Detail the matters to which you would pay particular attention in obtaining the required knowledge of the business and in developing your audit plan for the year ended 31 January 2013. (10 marks)

Study smarter with the SolutionInn App


