A 10 cm column of air is trapped by a column of Hg, 8 cm long,...
Fantastic news! We've Found the answer you've been seeking!
Question:
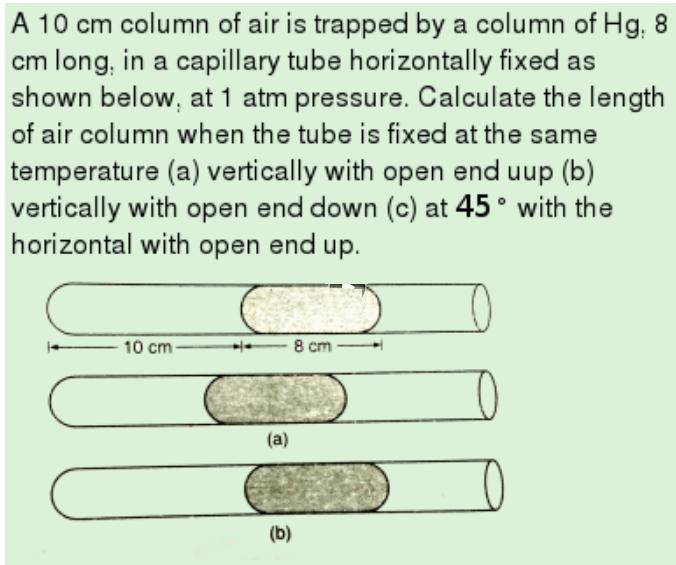
Transcribed Image Text:
A 10 cm column of air is trapped by a column of Hg, 8 cm long, in a capillary tube horizontally fixed as shown below, at 1 atm pressure. Calculate the length of air column when the tube is fixed at the same temperature (a) vertically with open end uup (b) vertically with open end down (c) at 45° with the horizontal with open end up. 10 cm 8 cm (a) (b) A 10 cm column of air is trapped by a column of Hg, 8 cm long, in a capillary tube horizontally fixed as shown below, at 1 atm pressure. Calculate the length of air column when the tube is fixed at the same temperature (a) vertically with open end uup (b) vertically with open end down (c) at 45° with the horizontal with open end up. 10 cm 8 cm (a) (b)
Expert Answer:
Answer rating: 100% (QA)
a at the horizontal position of tube air pressure is 1 atm mean... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
Propane gas is burned steadily at 1 atm pressure with a 10 percent excess oxygen supplied by atmospheric air. The reactants enter a steady flow combustor at 25C. Determine the final temperature of...
-
Water is boiled at 1 atm pressure in a 25-cm-internaldiameter stainless steel pan on an electric range. If it is observed that the water level in the pan drops by 10 cm in 45 min, determine the rate...
-
Water is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 2 kg of liquid water evaporates in 30 min. The rate of heat transfer to the water is (a) 2.51 kW...
-
Find dy/dx for the following functions. y COS X sin x + 1
-
The following reaction equation represents complete combustion of a fuel. Calculate the percent excess air. C2H6 1 6.5(O2 1 3.76N2) 1 2CO2 1 3H2 O 1 3O2 1 24.44N2 (A) 56% (B) 86% (C) 136% (D) 176%
-
The capacitor in FIGURE P28.73 begins to charge after the switch closes at t = 0 s. a. What is V C a very long time after the switch has closed?b. What is Q max in terms of , R, and C?c. In this...
-
James Lewis, a resident of Kentucky, sustained an injury while operating a Caterpillar bulldozer. He filed suit against Caterpillar, a company incorporated in Delaware but with its principal place of...
-
The following income statement was drawn from the records of Ulrich Company, a merchandising firm: .:. Required a. Reconstruct the income statement using the contribution margin format. b. Calculate...
-
Two years after the first round XMP GmbH receives a second offer form B-Capital. B-Capital offers to invest 12,500,000.00 at a 60,000,000.00 pre-money valuation. What is the founders' post-round...
-
Tony Anderson Productions is a sole trader-ship. On December 31, 2010 the following Trial Balance was extracted from the business's books: Trial Balance as at December 31, 2010 Details Return inwards...
-
1. How does an event planning company develop a consistent message about its services? How does it best generate buzz? 2. What approaches to Advertising, Sales Promotion, Social Media,...
-
Explain how the calculations were performed in SUN.SCR For example, were the numbers 93,000,000 and 186,000 represented anywhere? If not, why not?
-
You are the new CEO of CVS Pharmacies. Your goal is to create a healthier organizational culture. The first thing you do as CEO is implement an organizational culture that has the espoused values of...
-
A popular construct in 'artificial intelligence' is the recurrent neural network (RNN). RNNs are high-dimensional dynamical systems. Here is one possible model of a 'vanilla' RNN in continuous time:...
-
Financial statement analysis is an important assessment tool for businesses; however, what value does ratio analysis provide for budgeting in the public sector and why is it beneficial for a criminal...
-
Terry is a salaried nonexempt employee who earns annually $48,750 for a 35-hour workweek that is paid semimonthly. Terry is occasionally on-call for his employer. During the on-call time, Terry is...
-
Mention some Communications issues in a workplace. Mention the impact of generational differences and evolving technologies on organizational communications. What strategies could one employ in...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
Solve this system of three equations with three unknowns using EES: 2x - y + z = 7 3x2 + 3y = z + 3 xy + 2z = 4
-
Show that processes involving rapid chemical reactions are irreversible by considering the combustion of a natural gas (e.g., methane) and air mixture in a rigid container.
-
Repeat Prob. 14-131 for a total pressure of 90 kPa for air. Prob. 14-131 Air enters an air-conditioning system that uses refrigerant-134a at 30oC and 70 percent relative humidity at a rate of...
-
Demonstrate that both the Berry phase \(\gamma_{n}\) and the Berry curvature \(\boldsymbol{\Omega}_{n}(\boldsymbol{R})\) are invariant under a local gauge transformation.
-
Show that under a local gauge transformation (14.5) the vector potential A and the form of the wavefunction (28.7) are changed, but no observable is affected. Data from Eq. 28.7 Data from Eq. 14.5...
-
Evaluate the formula for the Gauss-Bonnet theorem in Box 28.3 for a 2-sphere and show that this leads to the usual relation for the area of a sphere. The local curvature for a 2-surface is the...

Study smarter with the SolutionInn App


