Question: Classify bond as ionic or covalent from formula. (Pool 1 of 2) Determine whether bonds in the following species are ionic or covalent Cl

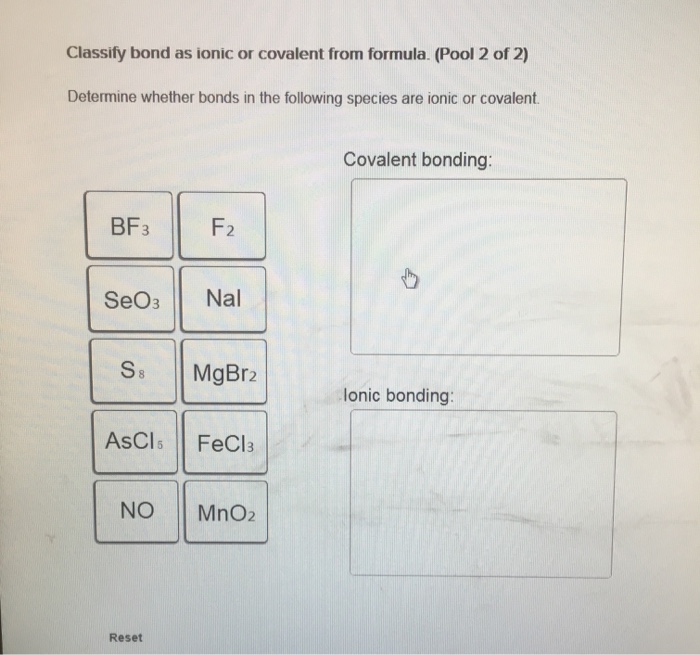

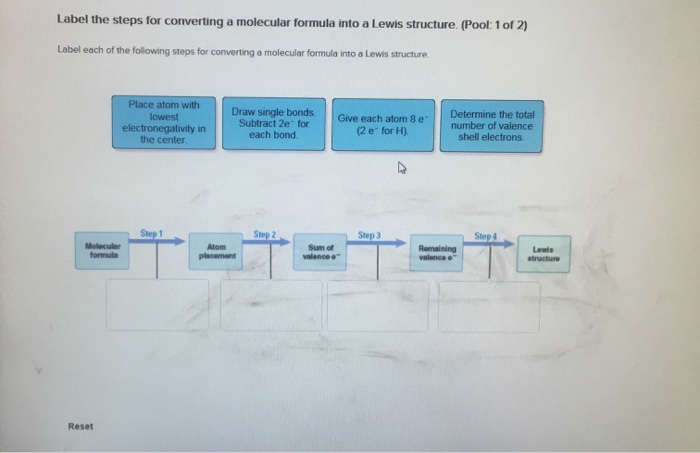

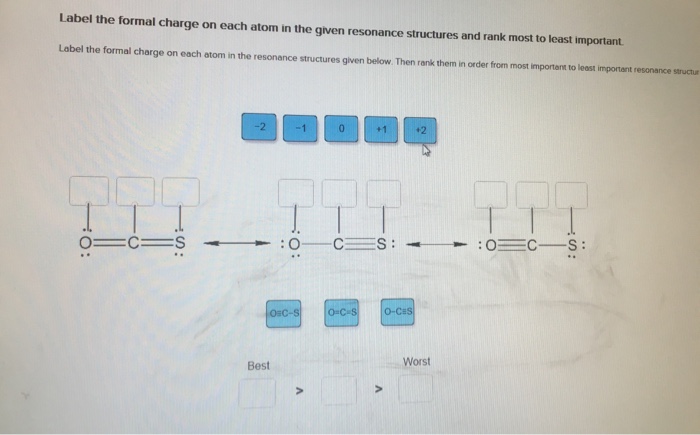
Classify bond as ionic or covalent from formula. (Pool 1 of 2) Determine whether bonds in the following species are ionic or covalent Cl CO CaF2 CrO2 HF Reset References Classification CaO P4 KCI SiCl4 CUS Covalent bonding: lonic bonding: Classify bond as ionic or covalent from formula. (Pool 2 of 2) Determine whether bonds in the following species are ionic or covalent. BF3 SeO3 S8 ASCI 5 NO Reset F2 Nal MgBr2 FeCl3 MnO2 Covalent bonding: D lonic bonding: Complete and sequence the steps for drawing Lewis structures. Complete and correctly sequence the steps for drawing Lewis structures. octet add multiple core lone surrounding four single six singles lower Reset pairs one bonding subtract central valence Zoom less eight more two higher wwwwwwwww AAAAAAAAAAAwww Drag the text blocks below into their correct order. Distribute the remaining electrons as up with electrons (or pairs on atoms to give each an the electrons If the make a Determine the total number of For polyatomic ions, charge, or from one of the the Draw a central atom, and bond. atom. Then check that each atom has so that each atom ends for H). First place electronegative) atom. If any electrons remain, place them on electrons available. one electron for each negative one electron for each positive charge. atom still does not have a(n) bond by changing a atoms into a bond from each surrounding atom to the two valence electrons for each Place the atoms relative to each other. Place the atom with number, place the atom with the the center. pair pair to group number in the center because it has the electronegativity. If atoms have the same group period number in Label the steps for converting a molecular formula into a Lewis structure. (Pool: 1 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Place atom with lowest electronegativity in the center. Molecular formula Reset Step 1 Draw single bonds. Subtract 2e for each bond. Atom placement Step 2 Sum of valence o Give each atom 8 e (2 e for H). Step 3 Determine the total number of valence shell electrons. Remaining valence Step 4 Lowie structure 14. value: 6.52 points Step 1 Label the steps for converting a molecular formula into a Lewis structure. (Pool: 2 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Remaining valence e < Place atom with lowest EN in center Question 14 (of 23) Sum of valence e Molecular formula Step 2 Total the number of valence shell electrons Lewis Structure Step 3 Draw single bonds, and subtract 20 for each bond Atom placement Step 4 Give each atom Be (2e" for H) Label the formal charge on each atom in the given resonance structures and rank most to least important. Label the formal charge on each atom in the resonance structures given below. Then rank them in order from most important to least important resonance structur 0=C=S -2 -1 Best OSC-S 0 > :0 CES: - +1 O-C-S A +2 O-CES Worst :0 CS:
Step by Step Solution
3.35 Rating (158 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


