Question: emergency: need answer asap Which has the higher boiling point? begin{tabular}{l|l} TheenthalpyofvaporizationofSubstanceAissmallerthanthatofSubstanceB. & Substance A & Substance B & Neither, A and B
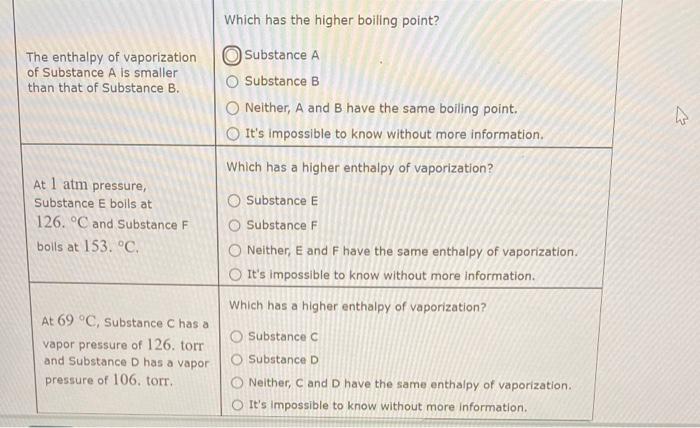
Which has the higher boiling point? \begin{tabular}{l|l} TheenthalpyofvaporizationofSubstanceAissmallerthanthatofSubstanceB. & Substance A \\ & Substance B \\ & Neither, A and B have the same boiling point. \end{tabular} It's impossible to know without more information. Which has a higher enthalpy of vaporization? At 1atm pressure, Substance E Substance E boils at Substance F Neither, E and F have the same enthalpy of vaporization. It's impossible to know without more information. Which has a higher enthalpy of vaporization
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


