Question: In Table C.2, at what reference temperature and pressure is the entropy zero? TABLE C.2 Thermodynamic Properties of Air at 1 atm* h (kJ/kg) u
In Table C.2, at what reference temperature and pressure is the entropy zero?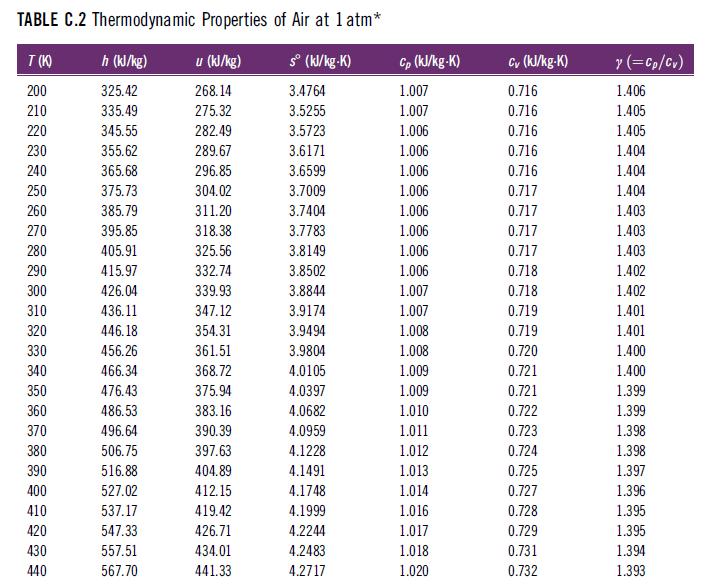
TABLE C.2 Thermodynamic Properties of Air at 1 atm* h (kJ/kg) u (kJ/kg) s (kJ/kg-K) 325.42 268.14 3.4764 335.49 275.32 3.5255 345.55 282.49 3.5723 355.62 289.67 3.6171 365.68 296.85 3.6599 375.73 304.02 3.7009 385.79 311.20 3.7404 395.85 318.38 3.7783 405.91 325.56 3.8149 415.97 332.74 3.8502 426.04 339.93 3.8844 436.11 347.12 3.9174 446.18 354.31 3.9494 456.26 361.51 3.9804 466.34 368.72 4.0105 476.43 375.94 4.0397 486.53 383.16 4.0682 496.64 4.0959 506.75 4.1228 516.88 4.1491 527.02 4.1748 537.17 4.1999 547.33 4.2244 557.51 4.2483 567.70 4.2717 T(K) 200 210 220 230 240 250 260 270 280 290 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 390.39 397.63 404.89 412.15 419.42 426.71 434.01 441.33 Cp (kJ/kg-K) 1.007 1.007 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.007 1.007 1.008 1.008 1.009 1.009 1.010 1.011 1.012 1.013 1.014 1.016 1.017 1.018 1.020 Cv (kJ/kg-K) 0.716 0.716 0.716 0.716 0.716 0.717 0.717 0.717 0.717 0.718 0.718 0.719 0.719 0.720 0.721 0.721 0.722 0.723 0.724 0.725 0.727 0.728 0.729 0.731 0.732 y (=Cp/Cv) 1.406 1.405 1.405 1.404 1.404 1.404 1.403 1.403 1.403 1.402 1.402 1.401 1.401 1.400 1.400 1.399 1.399 1.398 1.398 1.397 1.396 1.395 1.395 1.394 1.393
Step by Step Solution
3.30 Rating (156 Votes )
There are 3 Steps involved in it
Table C2 lists the specific enthalpy h and specific entropy s of various pure substances at differen... View full answer

Get step-by-step solutions from verified subject matter experts


