One kilogram of carbon dioxide is expanded adiabatically in a pistoncylinder system from 10 bar and 500
Question:
One kilogram of carbon dioxide is expanded adiabatically in a piston–cylinder system from 10 bar and 500 K to the saturated-vapor state at 5 bar. How much work is done by carbon dioxide and on what? Use the data in Appendix A.9.
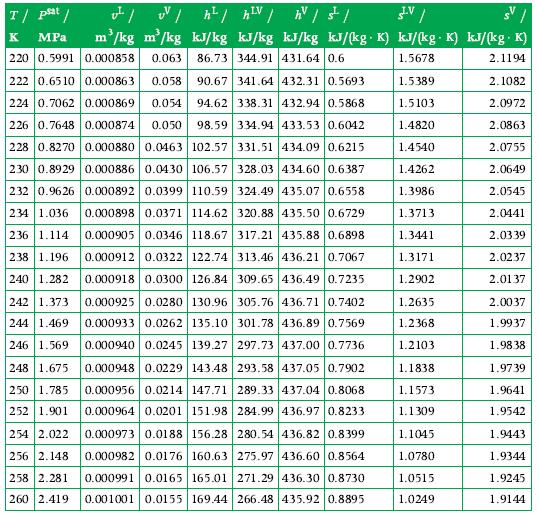
Transcribed Image Text:
T/ psat / K MPa 220 0.5991 222 0.6510 0.000863 0.058 90.67 341.64 432.31 0.5693 224 0.7062 0.000869 0.054 94.62 338.31 432.94 0.5868 226 0.7648 0.000874 0.050 98.59 334.94 433.53 0.6042 228 0.8270 0.000880 0.0463 102.57 331.51 434.09 0.6215 230 0.8929 0.000886 0.0430 106.57 328.03 434.60 0.6387 232 0.9626 0.000892 0.0399 110.59 324.49 435.07 0.6558 234 1.036 0.000898 0.0371 114.62 320.88 435.50 0.6729 236 1.114 0.000905 0.0346 118.67 317.21 435.88 0.6898 238 1.196 0.000912 0.0322 122.74 313.46 436.21 0.7067 240 1.282 0.000918 0.0300 126.84 309.65 436.49 0.7235 242 1.373 0.000925 0.0280 130.96 305.76 436.71 0.7402 244 1.469 0.000933 0.0262 135.10 301.78 436.89 0.7569 246 1.569 0.000940 0.0245 139.27 297.73 437.00 0.7736 248 1.675 0.000948 0.0229 143.48 293.58 437.05 0.7902 250 1.785 0.000956 0.0214 147.71 289.33 437.04 0.8068 252 1.901 0.000964 0.0201 151.98 284.99 436.97 0.8233 254 2.022 0.000973 0.0188 156.28 280.54 436.82 0.8399 256 2.148 0.000982 0.0176 160.63 275.97 436.60 0.8564 258 2.281 0.000991 0.0165 165.01 271.29 436.30 0.8730 260 2.419 0.001001 0.0155 169.44 266.48 435.92 0.8895 LV v²/w/h² | h²V / hv|s²| m³/kg m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg. K) kJ/(kg. K) kJ/(kg. K) 0.000858 0.063 86.73 344.91 431.64 0.6 1.5678 2.1194 1.5389 2.1082 1.5103 2.0972 1.4820 2.0863 1.4540 2.0755 1.4262 2.0649 1.3986 2.0545 1.3713 2.0441 1.3441 2.0339 1.3171 2.0237 1.2902 2.0137 1.2635 1.2368 1.2103 1.1838. 1.1573 1.1309 1.1045 1.0780 1.0515 1.0249 / 2.0037 1.9937 1.9838 1.9739 1.9641 1.9542 1.9443 1.9344 1.9245 1.9144
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The temperature of state 1 reported in ...View the full answer

Answered By

Tamil Elakkiya Rajendran
I'm currently involved in the research in the field of Biothermodynamics, Metabolic pathway analysis and computational Biology. I always prefer to share my knowledge whatever I have learnt through my degree whenever time permits.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
One kilogram of carbon dioxide is compressed from 0.5 MPa and 200oC to 3 MPa in a piston - cylinder device arranged to execute a polytropic process with n 5 1.3. Use the compressibility factor to...
-
Carbon dioxide sublimes at pressures below roughly 5 atm? At a pressure of 2 atm this phase transition occurs at about ?69 ? C with an enthalpy of sublimation of roughly 26 kJ/mol. Suppose a kilogram...
-
The two-year zero rate is 2% and the four-year zero rate is 3%. What is the forward rate for an investment starting at the end of the second year and maturing at the end of the fourth year? All rates...
-
As speed increases for an object in free fall, does acceleration increase also?
-
During the year just ended, Anna Schultzs portfolio, which has a beta of 0.90, earned a return of 8.6%. The risk-free rate is currently 7.3%, and the return on the market portfolio during the year...
-
A 2.0 mol sample of an ideal monatomic gas undergoe the reversible process shown in Figure, the scale of the vertical axis is set by Ts = 400.0K and the scale of the horizontal axis is set by Ss =...
-
Configure a work breakdown structure (WBS) based on scope and plan.
-
Ryans Express has total credit sales for the year of $180,000 and estimates that 3% of its credit sales will be uncollectible. Record the end-of-period adjusting entry on December 31, in general...
-
8 1 A liquid (p = 1.653) flows through a horizontal pipe of varying cross section. In the first section, the cross-sectional area is 10 cm2, the flow speed is 275 cm and the pressure is 1.20 x 105...
-
Calculate the mass flow rate of the steam that enters an adiabatic duct with a velocity of 10 m/s, at a pressure of 1.5 bar and a temperature of 400C. The duct has a diameter of 0.16 m at the inlet...
-
One kilogram of R134a is heated in a constant volume container from the critical point to 450 K. How much energy is transferred as heat to the fluid? Use the diagrams or data in Appendix A.4. Data...
-
Which approach to personality emphasizes the innate goodness of people and their desire to grow? a. Humanistic b. Psychodynamic c. Learning d. Biological and evolutionary
-
State the derivative of the following functions a) y = -x b) y= c) y = 2cos(x) d) y = 2* e) y = - f) y = ex
-
Evaluate the limit lim *-2 1-23-2-1
-
Bank Citibank EURUSD Spot Quotes Bid Ask 1.1930 1.1935 HSBC 1.1935 1.1940 JP Morgan Chase 1.1925 1.1930
-
International Law is one solution to the problem of peace. States, in their own interest, based on their own sovereignty, are able to undertake collective action and the combined efforts make...
-
Warnerwoods Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for March. Date March 1 March 5 March 9. March 18 Sales Activities Beginning...
-
Solid KSCN was added to a 2.00 M Co2+ solution so that it was also initially 2.00 M SCN. These ions then reacted to give the complex ion Co(SCN)+, whose formation constant was 1.0 102. What is the...
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
A system is made up of a vertical cylinder which is sealed at the top and closed by a piston at the bottom. A valve A controls the intake of gas at the top and an exhaust valve B (also at the top) is...
-
The surface tension modifies the melting point of particles. The effect is important when the diameter is in the nanometer range. A differential equation has to be written for Tm (r), the melting...
-
James Atkinson was a British engineer who designed several combustion engines. The thermodynamic cycle bearing his name is a modification of the Otto cycle intended to improve its efficiency. The...
-
nces Fogerty Company makes two products-titanium Hubs and Sprockets. Data regarding the two products follow: Direct Labor-Hours per Hubs Sprockets Unit 0.80 0.40 Annual Production 16,000 units 49,000...
-
Sandhill Water Co. is a leading producer of greenhouse irrigation systems. Currently, the company manufactures the timer unit used in each of its systems. Based on an annual production of 44,500...
-
EXHIBIT 5.10 Internal Control Questionnaire-Payroll Processing Control Environment 1. Are all employees paid by check or direct deposit? 2. Is a special payroll bank account used? 3. Are payroll...

Study smarter with the SolutionInn App


