Heat-transfer data are sometimes given as a j factor, where Show that such a correlation can be
Question:
Heat-transfer data are sometimes given as a j factor, where
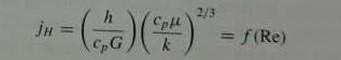
Show that such a correlation can be converted to one that gives the Nusselt number (Nu = hD/k) as a function of the Reynolds number (DG/μ) and the Prandtl number (cpμ/k).
Transcribed Image Text:
2/3 jH = ( ) () - ( = f (Re) CpG
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To convert a correlation given in terms of the j factor JH to one that gives the Nusselt number Nu a...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Unit Operations Of Chemical Engineering
ISBN: 9780072848236
7th Edition
Authors: Warren McCabe, Julian Smith, Peter Harriott
Question Posted:
Students also viewed these Engineering questions
-
Kintel, Inc., management wants to raise $1 million by issuing six-year zero coupon bonds with a face value of $1,000. The companys investment banker states that investors would use an 8.9 percent...
-
The site http://virtualpiano.net features an online player piano. If you click on the Key Assist menu just above the piano keyboard, and then Keyboard Keys, you will see that each key is labelled...
-
Sure, let's go step by step: **(1) Open a general ledger account for each account in the chart of accounts:** Here's a simplified representation: ``` ASSETS 101 Cash in Bank 105 Accounts...
-
Pohle Designs has a department that makes high-quality leather cases for iPads. Consider the following data for a recent month: Budget Formula per Unit Various Levels of Output 10.000 1.000 12000...
-
a. Formulate a linear programming model for the Mosaic Tile Company to determine the mix of the tiles it should manufacture each week. b. Transform the model into standard form. c. Solve the linear...
-
The Ombudsman Foundation is a private not-for-profit organization providing training in dispute resolution and conflict management. The Foundation had the following pre-closing trial balance at...
-
Suppose there are \(n\) stocks. Each of them has a price that is governed by geometric Brownian motion. Each has \(v_{i}=15 \%\) and \(\sigma_{i}=40 \%\). However, these stocks are correlated, and...
-
Prepare the journal entry to record Typist Companys issuance of 250,000 shares of its common stock assuming the shares have a: a. $1 par value and sell for $10 cash per share. b. $1 stated value and...
-
The following year-end information is taken from the December 31 adjusted trial balance and other records of Leone Company. Advertising expense Depreciation expense-Office equipment $ 28,750 7,250...
-
In many countries in South America, chunks of cassava root containing 65 percent water are dried to 5 percent water, then ground to make tapioca flour. To make 1,200 kg/h of flour, what would be the...
-
At what point is the temperature in degrees Fahrenheit equal to the Celsius temperature? Is there any point where the Kelvin temperature is the same as the Rankine temperature?
-
How can it be fair for Bakie to owe double the value of the tractor when it no longer even has the machine?
-
A 0.475-kg hockey puck, moving east with a speed of 2.00 m/s, has a head-on collision with a 0.850- kg puck initially at rest Assuming a perfectly elastic collision, what will be the speed (magnitude...
-
discuss the principles of immunological tolerance and self-tolerance mechanisms, including central tolerance mechanisms in the thymus and bone marrow, and peripheral tolerance mechanisms such as...
-
Can you elucidate the molecular mechanisms underlying antigen presentation by major histocompatibility complex (MHC) molecules, including the roles of MHC class I and II molecules in presenting...
-
What are the main barriers to adopting investment appraisal techniques in business? In what ways can these problems be overcome in part/full?
-
What did you learn? (For example, consider what you may have learned about criminal law, the concept of crime, and criminal liability.) What surprised you? What impact did the interview have on your...
-
A randomized block experiment produced the following statistics: k = 5 b = 12 SST = 1,500 SSB = 1,000 SS(Total) = 3,500 a. Test to determine whether the treatment means differ. (Use = .01.) b. Test...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
Succinic acid dissociates in two steps: K1 H,H,C ,, + H* %3| || OCCH,CH,CO + H* , 3 2.3 10-6 HOCCH,CH,CO Calculate Kp1 and Kp2 for the following reactions: || OCCH,CH,CO + H,0 = HOCCH,CH,CO + OH ...
-
Histidine is a triprotic amino acid: What is the value of the equilibrium constant for the reaction CO,H -NH CO, -NH HC-CH, K, = 3 x 102 HC-CH, NH, NH, , 3 8.5 10-7 CO, -NH CO NH HC-CH2 , 3 4.6 10...
-
(a) From Kw in Table 6-1, calculate the pH of pure water at 0, 20, and 40C. (b) For the reaction D 2 O D + + OD - , K = [D + ][OD - ] =1.35 10 -15 at 25C. In this equation, D stands for deuterium,...
-
11. Given f(x) = 7x-2, find f(x+3)+f(-1)
-
In 1999, GLSEN identified that little was known about the school experiences of lesbian, gay, bisexual, transgender, and queer (LGBTQ+) youth and that LGBTQ+ youth were nearly absent from national...
-
1. In each of the following cases, draw a graph showing an increase or decrease in equilibrium interest rates. a. Suppose the demand for loanable funds remains unchanged. Will the equilibrium...

Study smarter with the SolutionInn App


