Question: Repeat Problem 4.48 using Fig. 4.48, except that there is now 4.65 kPa air pressure above the fluid. Ammonia sg = 0.826 0,62 m 1.25
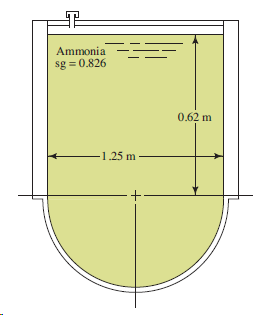
Ammonia sg = 0.826 0,62 m 1.25 m
Step by Step Solution
3.34 Rating (154 Votes )
There are 3 Steps involved in it
h 1e h 1 h a 062 m 0574 m 1194 m ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
1822_604739cc0b5f4_724324.xlsx
300 KBs Excel File


