Calculate the standard heat of reaction for the following reaction: the hydrogenation of benzene to cyclohexane. (1)
Question:
Calculate the standard heat of reaction for the following reaction: the hydrogenation of benzene to cyclohexane.
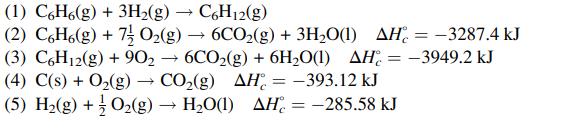
Transcribed Image Text:
(1) C6H6(g) + 3H₂(g) → C6H12(g) (2) C6H6(g) + 7 O₂(g) 6CO₂(g) + 3H₂O(1) AH-3287.4 kJ (3) C6H12(g) + 90₂ → 6CO₂(g) + 6H₂O(1) AH-3949.2 kJ CO₂(g) AH = -393.12 kJ (4) C(s) + O₂(g) (5) H₂(g) + O₂(g) H₂O(1) AH= -285.58 kJ → -
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Method 1 Using the more general equation 326 ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
All chemists know that benzene is unusually stable, that is, it is aromatic. They are also well aware that many other similar molecules are stabilized by aromaticity to some extent and, more often...
-
Calcium chloride is a salt used in a number of food and medicinal applications and in brine for refrigeration systems. Its most distinctive property is its affinity for water: in its anhydrous form...
-
The standard heat of combustion of gaseous acetylene is listed in Table B.I as 1299.6kJ/mol. (a) In your own words, briefly explain what that means. (Your explanation should mention the reference...
-
Rainfall of magnitude 3.8cm and 2.8cm occurring on two consecutive 4-h durations on a catchment of area 27km produced the following hydrograph of flow at the outlet of the catchment. Estimate the...
-
The following summary data are from the May 31, 2004, balance sheet of FedEx. All numbers are in millions. Total current assets. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 4,970...
-
Identify the three phases of the auditing process and indicate the time frame when each is performed.
-
True or False. A random variable is a quantity whose magnitude cannot be predicated precisely.
-
A golf specialty wholesaler operates 50 weeks per year. Management is trying to determine an inventory policy for its 1-irons, which have the following characteristics: Demand (D) = 2,000 units/year....
-
The terms below are just a few terms needed in discussing the world of social media marketing and it is important that students have a clear and consistent understanding of these terms. We will use...
-
Calculate the liquidation of the Corporate Tax in a word document, using the following scheme.: Accounting result +/- Permanent differences +/- Temporal differences - Set-off for tax bases from...
-
Illustrates the manual calculation of a reactor heat balance. Vinyl chloride (VC) is manufactured by the pyrolysis of 1,2-dichloroethane (DCE). The reaction is endothermic. The flow-rates to produce...
-
Methane is compressed from 1 bar and 290 K to 10 bar. If the isentropic efficiency is 0.85, calculate the energy required to compress 10,000 kg/h. Estimate the exit gas temperature.
-
For the first-order reaction t 1/2 = 22.5 h at 20 C and 1.5 h at 40 C. (a) Calculate the activation energy of this reaction. (b) If the Arrhenius constant A = 2.05 x 10 13 s -1 , determine the value...
-
This week we learned about the courts, their operations and the different parties in litigation. Please use course, library and online means to find academic quality resources, then answer the...
-
What if you are in the car with a friend when you are pulled over by police and the officer draws his gun and tells you and your friend to get out of the car? You know you have done nothing wrong....
-
What is the central idea of this article. Howe, Scott W. (2015) "The Implications of Incorporating the Eighth Amendment Prohibition on Excessive Bail," Hofstra Law Review: Vol. 43: Iss. 4, Article 4....
-
Richard Delgado, "Storytelling for Oppositionists and Others: A Plea for Narrative" Michigan Law Review 87 (1989): 2411-2441. LINK...
-
What would be the consequence of implementing maximum punishment (death penalty) for all crimes, regardless of how severe they are? Would the crime rate increase or decrease? Would they become more...
-
Why have FIs been very active in loan securitization issuance of pass-through securities while they have reduced their volume of loan sales? Under what circumstances would you expect loan sales to...
-
Thalina Mineral Works is one of the worlds leading producers of cultured pearls. The companys condensed statement of cash flows for the years 20182020 follows. Required Comment on Thalina Mineral...
-
Refer to the Fourier transform infrared spectrum in Figure 19-32. (a) The interferogram was sampled at retardation intervals of 1.2660 10 -4 cm. What is the theoretical wavenumber range (0 to ?) of...
-
The table shows signal-to-noise ratios recorded in a nuclear magnetic resonance experiment. Construct graphs of (a) signal-tonoise ratio versus n and (b) signal-to-noise ratio versus n, where n is...
-
Would you use a tungsten or a deuterium lamp as a source of 300-nm radiation? What kind of lamp provides radiation at 4-m wavelength?
-
Bond X is a premium $1000 par value bond making annual payments. The bond has a coupon rate of 9%, a YTM of 7%, and has 13 years to maturity. Bond Y is a discount $1000 par value bond making annual...
-
The growth rate for the firm's common stock is 7%. The firm's preferred stock is paying an annual dividend of $5. What is the preferred stock price if the required rate of return is 8%?
-
What do we know about the Galaxies over time according to https://webb.nasa.gov/content/science/galaxies.html What are some of the unknowns How might the JWT shed light on the galaxies over time?...

Study smarter with the SolutionInn App


