Nitrogen monoxide reacts with ozone. NO(g) + O3(g) NO(g) + O(g) Two mechanisms have been proposed: Mechanism
Question:
Nitrogen monoxide reacts with ozone.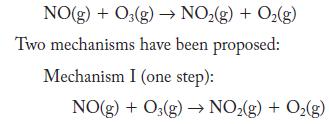
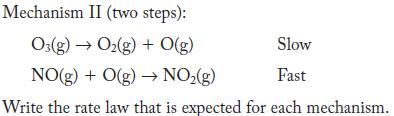
Transcribed Image Text:
NO(g) + O3(g) NO(g) + O(g) Two mechanisms have been proposed: Mechanism I (one step): NO(g) + O3(g) NO(g) + O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The rate law for a chemical reaction describes how the rate of reaction depends on the concentration ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Nitrogen monoxide reacts with oxygen to give nitrogen dioxide. 2NO(g)+O2(g) 2NO2(g) The rate law is [NO]/ t = k[NO]2[O2], where the rate constant is 1.16 105 L2/(mol2s) at 339oC. A vessel contains...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Consider the reaction of ozone and nitrogen monoxide to form nitrogen dioxide and oxygen. Which of the following orientations for the collision between ozone and nitrogen monoxide could perhaps lead...
-
Four of Wands, LLC generated $255,000 in sales during January 2022. Of this amount, 25% was for cash. The remaining 75% of sales were made on account. The February 2022 sales on account were...
-
Find I0 and I1 in the circuit infigure. 5 mA 4 mA 2 mA 3 mA ww (+1)
-
What important decision must be made when test of controls auditing is performed and control risk is evaluated at an interim date several weeks or months before the clients fiscal year-end?
-
Table B. 15 presents data on air pollution and mortality. Use the all-possibleregressions selection on the air pollution data to find appropriate models for these data. Perform a thorough analysis of...
-
Mario, age 65, purchased an immediate annuity for $120,000 that pays a lifetime monthly income of $1000. The annuity has no refund feature. Based on the IRS actuarial table, Mario has a life...
-
Suppose a 20-year Treasury bond with 5% semiannual coupon is currently traded at a yield of 6% (APR). 1) Is the price above or below 100? 2) What is the current price of the bond? 3) If the yield...
-
The gas-phase reaction of nitrogen monoxide with chlorine proceeds to form nitrosyl chloride. 2NO(g) + Cl(g) 2NOC1(g) rate = k[NO][C1] Evaluate the following proposed mechanism to determine whether...
-
In 1926, Hinshelwood and Green studied the reaction of nitrogen monoxide and hydrogen. (a) What is the rate law for the reaction? (b)Use the data from the first experiment to calculate the rate...
-
Are you required to use all five digits of a random number when you have a random number table, such as the one at the end of this appendix?
-
Job satisfaction has been defined in various ways. Some would argue it is simply how content a person is with his or her job. For this discussion, adhere to the FEM guidelines as you complete the...
-
Define the concept of Industrial-Organizational Psychology, along with its utilization in the workplace. (Important Note: neither of these tactics/strategies should be related to compensation; the...
-
What are the basic activities involved in the management process? Please explain. Describe the different types of managers and their roles within organizations. How is this related to their...
-
Many feel that the world's biggest retailer Walmart views Amazon as their biggest competitor/threat. In reading the Gartner Top 25 information on Amazon and doing some additional research on Amazon's...
-
As a manager, what are some ways you help increase job satisfaction in your employees? What are the organizational consequences when there are high or low levels of job satisfaction?
-
The Thompson Corporation projects an increase in sales from $18 million to $25 million, but it needs an additional $500,000 of current assets to support this expansion. Thompson purchases under terms...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Sketch the influence line for (a) The moment at E, (b) The reaction at C, and (c) The shear at E. In each case, indicate on a sketch of the beam where a uniform distributed live load should be placed...
-
Draw the influence line for the reaction at C. Plot the numerical values every 5 ft. EI is constant. B -15 ft- -15 ft-
-
Draw the influence line for the shear at C. Plot numerical values every 1.5 m. Assume A is fixed and the support at Bis a roller. EI is constant. SA B 3 m 3 m
-
In a study of factors that influence brand switching by a certain demographic, the following factors were identified as being significant as judged by the responses of the 439 participants in the...
-
When reviewing a budget comparison report, what can the reader determine?
-
Should I include the financial ratio information in this document in the plan to the board of directors?

Study smarter with the SolutionInn App


