A mixture of 1.00 mol NaHCO 3 (s) and 1.00 mol Na 2 CO 3 (s) is
Question:
A mixture of 1.00 mol NaHCO3(s) and 1.00 mol Na2CO3(s) is introduced into a 2.50 L flask in which the partial pressure of CO2 is 2.10 atm and that of H2O(g) is 715 mmHg. When equilibrium is established at 100 °C, will the partial pressures of CO2(g) and H2O(g) be greater or less than their initial partial pressures? Explain.
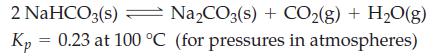
Transcribed Image Text:
2 NaHCO3(s) → Na₂CO3(s) + CO₂(g) + H₂O(g) 0.23 at 100 °C (for pressures in atmospheres) Kp
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To determine whether the partial pressures of CO2g and H2Og will be greater or less than their initi...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A 25-g block of iron at 175C is dropped into a liter of water in an insulated flask at 20C and 1 atm. The specific enthalpy of iron is given by the expression H(J/g)= 17.3T(C). (a) What reference...
-
In chemical vapor deposition (CVD), a semiconducting or insulating solid material is formed in a reaction between a gaseous species and a species adsorbed on the surface of silicon wafers (disks...
-
A mixture of 1 mol of H2O, 2 mol of O2, and 5 mol of N2 is heated to 2200 K at a pressure of 5 atm. Assuming the equilibrium mixture consists of H2O, O2, N2, and H2, determine the equilibrium...
-
Logical fallacies are frequently used in arguments and have an intuitive appeal that makes them effective for politicians to use. Using your understanding of fallacies, you will listen to and take...
-
Southland Industries has $60,000 of 16% (annual interest) bonds outstanding, 1,500 shares of preferred stock paying an annual dividend of $5 per share, and 4,000 shares of common stock outstanding....
-
On May 20, cattle rancher Oliver visited his neighbor Southworth, telling him, I know youre interested in buying the land Im selling. Southworth replied, Yes, I do want to buy that land, especially...
-
In spring 1989, Michael Jordan and the Chicago Bulls were in Indianapolis, Indiana, to play against the Indiana Pacers. At the same time, Karla Knafel was singing with a band at a hotel in...
-
Marwick's Pianos, Inc., purchases pianos from a large manufacturer and sells them at the retail level. The pianos cost, on the average, $2,450 each from the manufacturer. Marwick's Pianos, Inc.,...
-
A seal dives below the ice in search of fish. Its path can be modelled by where d is the depth in decameters and t the time in minutes. Use the equation above to answer questions about the seal's...
-
Cadmium metal is added to 0.350 L of an aqueous solution in which [Cr 3+ ] = 1.00 M. What are the concentrations of the different ionic species at equilibrium? What is the minimum mass of cadmium...
-
Formamide, used in the manufacture of pharmaceuticals, dyes, and agricultural chemicals, decomposes at high temperatures. If 0.186 mol HCONH 2 (g) dissociates in a 2.16 L flask at 400 K, what will be...
-
1. How might different types of office design influence employee social interaction, collaboration, and creativity? Should these be encouraged even in organizations without an innovative culture? 2....
-
Computer Geeks has sales of $566980, a profit margin of 0.53, a total asset turnover rate of 1.13, and an equity multiplier of 1.39. What is the return on equity?
-
Find the required rate of return for equity investors of a firm with a beta of 1.3 when the risk free rate is 5%, and the return on the market is 10%
-
For the steel beam and loading shown above (A-hinge support, C= Cable) (A) Write the equations for bending moment and shear force at any section along the beam (1 Point) and draw the shear force and...
-
Conduct research on and discuss new trends and research in parallel processing.
-
A stock just paid a dividend of D 0 = $1.50. The required rate of return is r s = 15.5%, and the constant growth rate is g = 4.0%. What is the current stock price?
-
Anita Theater is located in the Zurbrugg Mall. A cashiers booth is located near the entrance to the theater. Two cashiers are employed. One works from 15 P.M., the other from 59 P.M. Each cashier is...
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
The following are inputs and outputs to the copying process of a copy shop: Percent jobs done on time Number of times paper supply runs out Number of pages copied per hour Number of employee errors...
-
Bavarian Chocolate Company produces chocolate bars. The primary materials used in producing chocolate bars are cocoa, sugar, and milk. The standard costs for a batch of chocolate (5,000 bars) are as...
-
Hickory Furniture Company manufactures unfinished oak furniture. Hickory uses a standard cost system. The direct labor, direct materials, and factory overhead standards for an unfinished dining room...
-
What can 'netcat' be used for when trying to evade IDS?
-
Give an example of 4 entirely different possible investments that in your opinion are almost equally risky (on their own), have almost equal 'expected returns', but where the risks are highly...
-
In your own construct an overview of the critical infrastructure Information Technology sector. Part 2. Examine at least three of the following: Unique aspects as they relate to national security....

Study smarter with the SolutionInn App


