Question: Use data from Table 7.2 and r H for the following reaction to determine the standard enthalpy of formation of CCl 4 (g) at
Use data from Table 7.2 and ΔrH° for the following reaction to determine the standard enthalpy of formation of CCl4(g) at 25 °C and 1 bar.
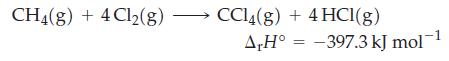
Table 7.2
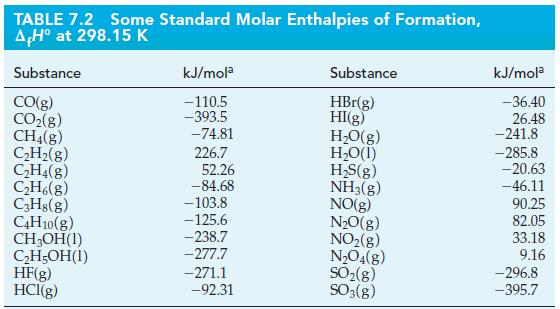
CH4(g) + 4Cl(g) CCl4(g) + 4HCI(g) AH -397.3 kJ mol
Step by Step Solution
3.43 Rating (150 Votes )
There are 3 Steps involved in it
To determine the standard enthalpy of formation of CCl4g at 25 C and ... View full answer

Get step-by-step solutions from verified subject matter experts


