Question: Use data from Table 7.3 and Appendix D to determine the standard enthalpy change in the following reaction. Table 7.3 Al+ (aq) + 3OH(aq) Al(OH)3(s)
Use data from Table 7.3 and Appendix D to determine the standard enthalpy change in the following reaction.
![]()
Table 7.3
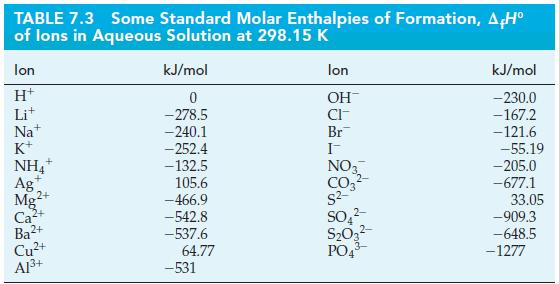
Al+ (aq) + 3OH(aq) Al(OH)3(s) A,H = ?
Step by Step Solution
3.36 Rating (159 Votes )
There are 3 Steps involved in it
Al30Haq AlOH36 for Al 531 KJmol AH for OHa... View full answer

Get step-by-step solutions from verified subject matter experts


