Suggest structures for compounds with the following proton NMR spectra. (a) CH0O: 8 1.13 (3H, t, J
Question:
Suggest structures for compounds with the following proton NMR spectra.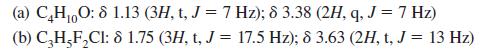
Transcribed Image Text:
(a) C₂H₁0O: 8 1.13 (3H, t, J = 7 Hz); 8 3.38 (2H, q, J = 7 Hz) 10 (b) C₂H5F₂Cl: 8 1.75 (3H, t, J = 17.5 Hz); 8 3.63 (2H, t, J = 13 Hz)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a Nothing but ethyls and one oxygen This can only be diethyl ether CH3C...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2 ? ?IR: 1735 cm ?1 ? (b) C 7 H 12 O 4 ? ? ?IR: 1735 cm ?1 ? (c) C 11 H 12 O 2 ? ??IR: 1710 cm ?1 ? TMS 8. 6. O ppm...
-
Propose structures for compounds with the following formulas that show only one peak in their 1H NMR spectra: (a) C5H12 (b) C5H10 (c) C4H8O2
-
Explain how the proton NMR spectra of the compounds within each of the following sets would differ, if at all. (CH)2CHC and (CH CDCI
-
The management of a New York area investment firm wants to find out about the investment needs of its existing customers, for which it has an extensively detailed list, as a function of their...
-
Rainey Company sells coffee makers used in business offices. Its beginning inventory of coffee makers was 200 units at $25 per unit. During the year, Rainey made two batch purchases of coffee makers....
-
Chris Guthrie was recently hired by S & S Air, Inc., to assist the company with its short-term financial planning and to evaluate the companys performance. Chris graduated from college five years ago...
-
A person donates a bag of clothes to Goodwill completely unaware that there is valuable sterling silver in the bag. The clothing and silver are subsequently sold, for a very economical price, to...
-
A liquid stream consisting of 12.5 mole% n-butane and the balance a heavy nonvolatile hydrocarbon is fed to the top of a stripping column, where it is contacted with an upward-flowing stream of...
-
A 2 kg block is attached to a spring with a force constant of 400 N/m. The block is initially at rest and is compressed by 0.5 meters from its equilibrium position. When released, the block undergoes...
-
Suppose you were able to cool a sample of 1-bromo-1,1,2-trichloroethane enough that rotation about the carboncarbon bond becomes slow on the NMR time scale. What changes in the NMR spectrum would you...
-
Explain how the NMR spectra of (a) 3-methyl-2-buten-1-ol and (b) 1,2,2-trimethyl-1-propanol would change following a D 2 O shake.
-
In 1981, Daniel Goodwin climbed 443 m up the exterior of the Sears Building in Chicago using suction cups and metal clips. (a) Approximate his mass and then compute how much energy he had to transfer...
-
Al tells Marge an offensive joke, but when Marge says Al, I dont appreciate your nasty jokes, Al responds, Im sorry, Marge, youre right, I shouldnt have told that one at work.
-
Describe how selection and training are related.
-
A male has asked two female co-workers to stop embarrassing him by telling jokes of a sexual nature and sharing their sexual fantasies, but they continue, telling him a real man wouldnt be...
-
Why is cultural training critical for employees embarking on an overseas assignment?
-
A female requests that her male assistant stay in her hotel room to save on expenses while out of town at a conference and holds acceptance as a job condition for continued employment.
-
Yung is the sole owner of Southern Hills Insurance Agency. His primary business is the sale of fire and casualty policies. He has recently expanded his business by selling life insurance policies....
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
One step in the urea cycle for ridding the body of ammonia is the conversion of arginino succinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the...
-
Although anti periplanar geometry is preferred for E2 reactions, it isn?t absolutely necessary. The deuterated bromo compound shown here reacts with strong base to yield an un-deuterated alkene....
-
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy? (a) CI...
-
ABC Company discovered an inventory error in its records. The company's physical count of inventory revealed a quantity of 500 units, while the recorded quantity in the books was 450 units. The cost...
-
In 2018 Larry's taxable income is $51,000, he received $550 in dividends from a Canadian-controlled private corporation. Using the federal tax rates below, how much net federal tax will Larry pay on...
-
Why do you think Domestic Violence is so prevalent in our communities? Please respond with a few sentences indicating why you think Domestic Violence is so prevalent in Orange County Communities. DO...

Study smarter with the SolutionInn App


