Question: The temperature increases from 300 K to 1200 K as a gas undergoes the process shown in FIGURE Q18.11. What is the final pressure? p
The temperature increases from 300 K to 1200 K as a gas undergoes the process shown in FIGURE Q18.11. What is the final pressure?
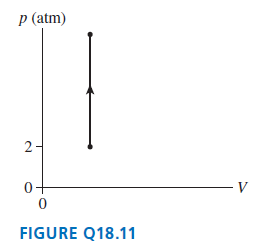
p (atm) 2- 0+ FIGURE Q18.11
Step by Step Solution
3.33 Rating (171 Votes )
There are 3 Steps involved in it
The figure shows that V 2 V ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
1442_6054778b9078d_693322.pdf
180 KBs PDF File
1442_6054778b9078d_693322.docx
120 KBs Word File


