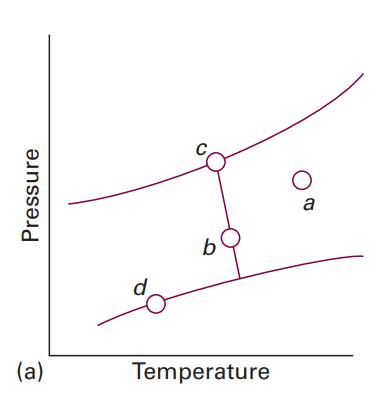
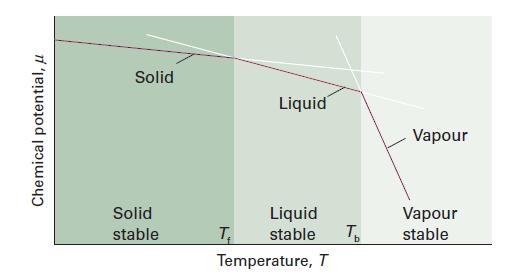
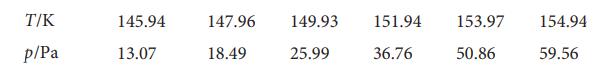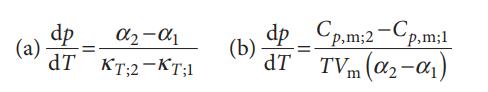Physical Chemistry 8th Edition Peter Atkins, Julio De Paula - Solutions
Unlock the secrets of "Physical Chemistry 8th Edition" by Peter Atkins and Julio De Paula with our comprehensive solutions manual. Dive into a world of online answers and find the answers key you need for your studies. Our solutions PDF provides step-by-step answers to solved problems, ensuring you grasp each concept thoroughly. Explore the test bank and instructor manual for a deeper understanding of chapter solutions, tailored for both students and instructors. With our textbook resources, questions and answers are just a click away, offering a seamless learning experience. Enjoy the convenience of free download options and enhance your knowledge with ease.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()