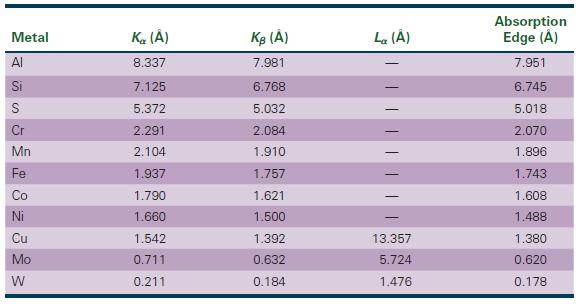
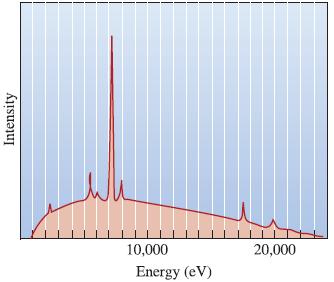
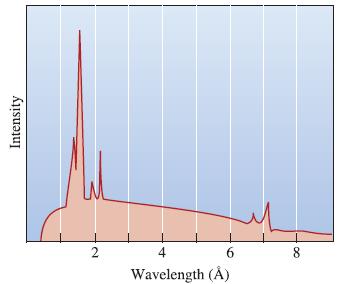
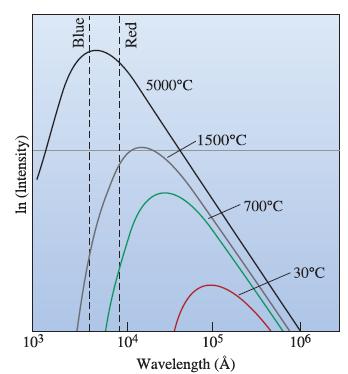
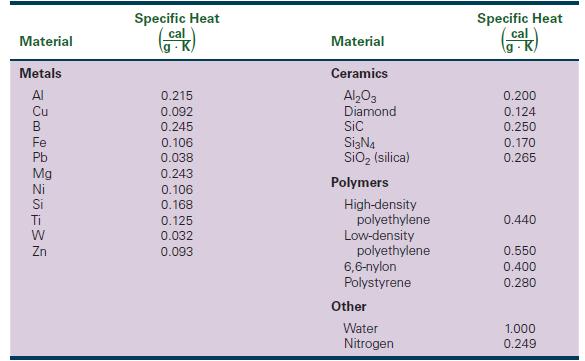
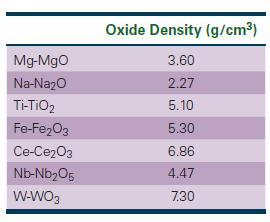
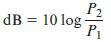The Science And Engineering Of Materials 7th Edition Donald R. Askeland, Wendelin J. Wright - Solutions
Unlock the full potential of "The Science And Engineering Of Materials 7th Edition" by Donald R. Askeland and Wendelin J. Wright with our comprehensive solutions. Discover detailed answers key and solutions manual available online, offering solutions PDF for all your queries. Dive into solved problems, and explore questions and answers that enhance your understanding. Our test bank and chapter solutions provide step-by-step answers, aligning perfectly with the instructor manual. Whether you're a student or educator, gain insights from this textbook with free download options, ensuring you stay ahead in your studies.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()