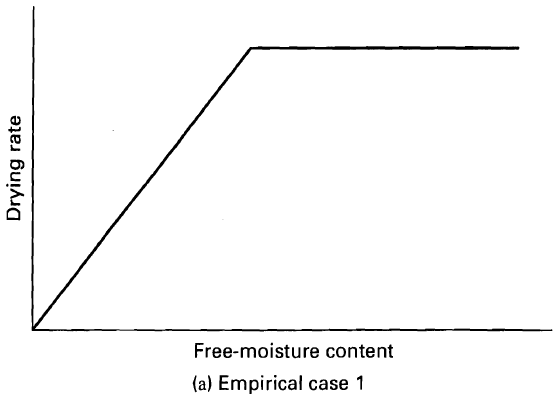
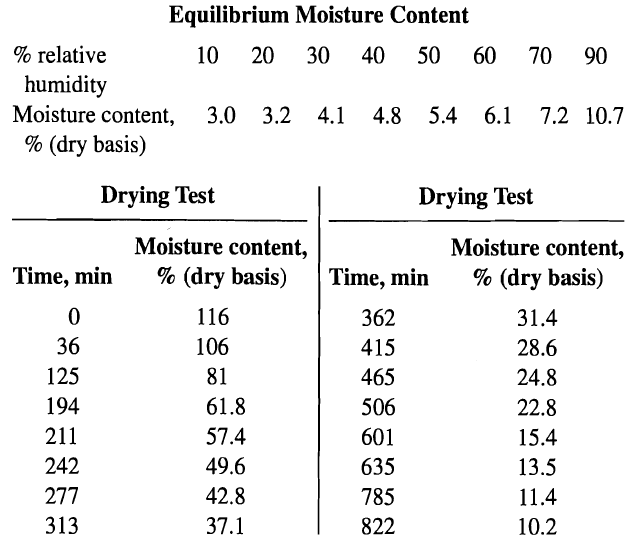
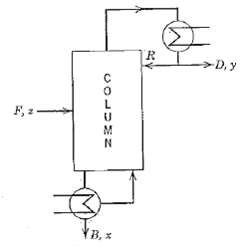
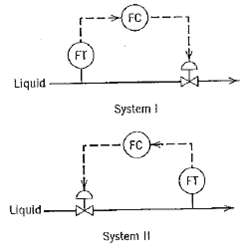
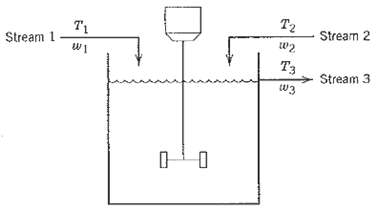
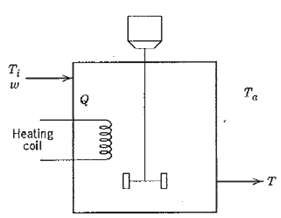
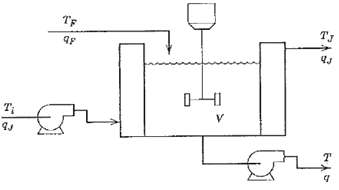
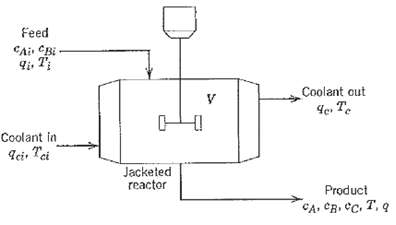
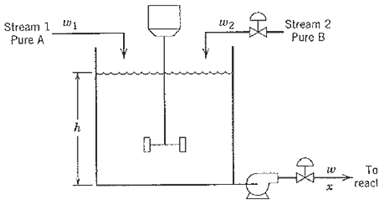
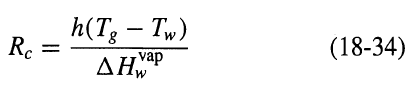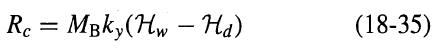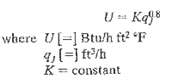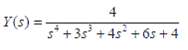Process Dynamics And Control 2nd Edition Dale E. Seborg, Thomas F. Edgar, Duncan A. Mellich - Solutions
Unlock comprehensive understanding with our expertly crafted solutions for "Process Dynamics And Control 2nd Edition" by Dale E. Seborg, Thomas F. Edgar, and Duncan A. Mellich. Access our online answers key, featuring detailed solutions pdf and a solution manual that breaks down complex concepts into manageable steps. Delve into solved problems with our step-by-step answers, ensuring clarity and comprehension for each chapter. Enhance your learning with our test bank, instructor manual, and textbook resources, all available for free download. Explore our questions and answers to navigate your academic journey with confidence.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()




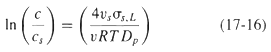

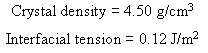
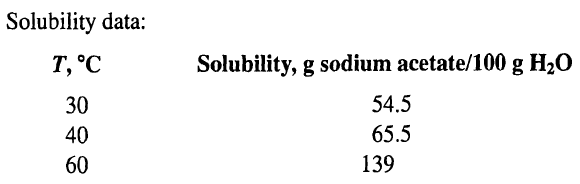
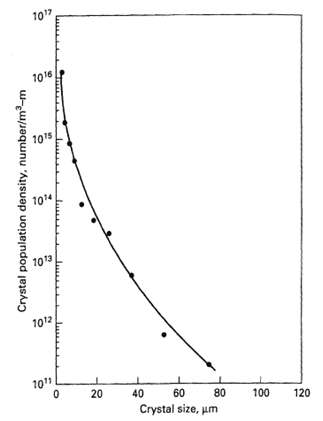
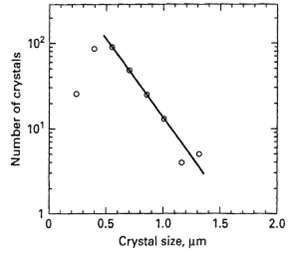
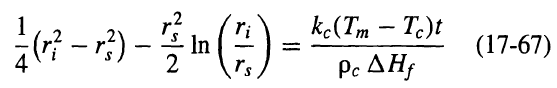
![e(1 - K) K(z2 – Z1) -lеxp( - 22 K /е) — ехp( -21 К /0)] + 1 W avg = Wo](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1554/2/7/2/3025ca4502e11a881554272304397.jpg)