Chemical Principles 7th edition Steven S. Zumdahl, Donald J. DeCoste - Solutions
Discover comprehensive solutions for "Chemical Principles 7th Edition" by Steven S. Zumdahl and Donald J. DeCoste with our expertly crafted answers key. Access detailed solutions manual and solutions PDF to tackle challenging problems with confidence. Our test bank and chapter solutions provide step-by-step answers to enhance your understanding of textbook concepts. Ideal for instructors and students, the instructor manual offers in-depth insights, while solved problems aid in mastering complex topics. Benefit from free download options to effortlessly integrate these resources into your study routine. Unlock success with our online questions and answers assistance today!
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()




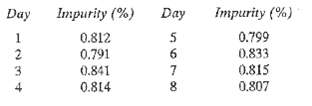


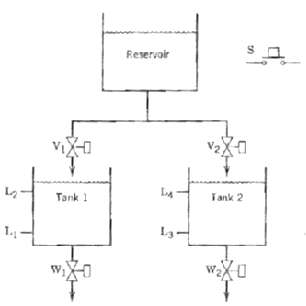
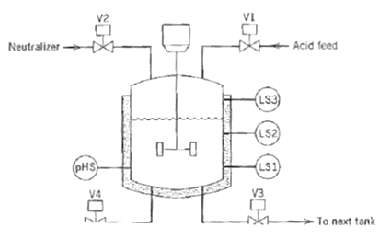
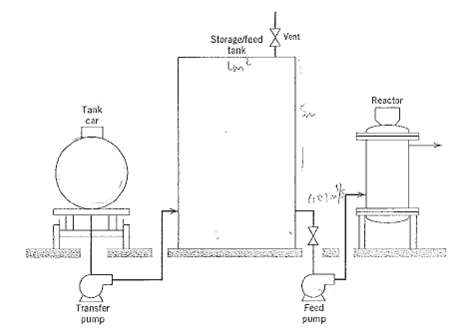
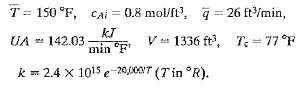
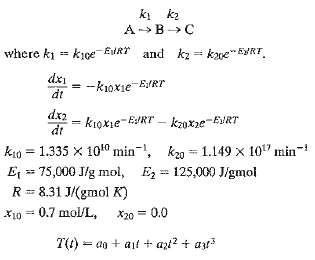
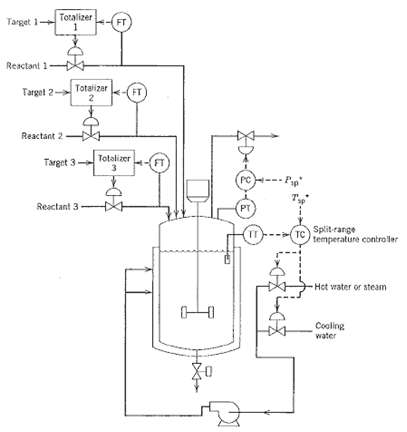
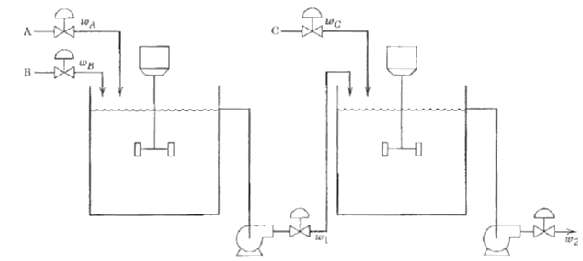

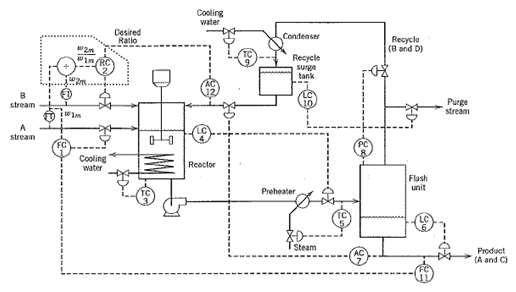
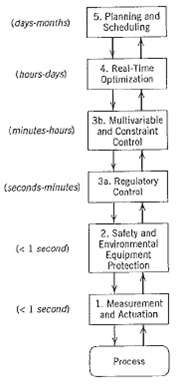
.png)
.png)

.png)
.png)
.png)