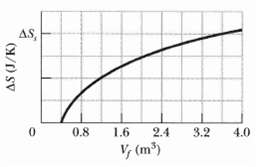
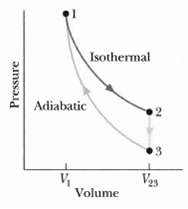
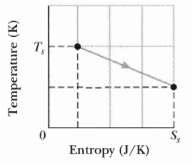
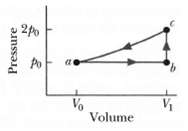
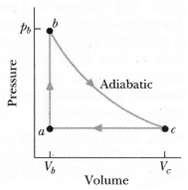
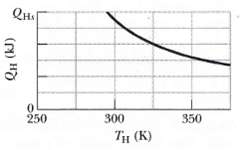
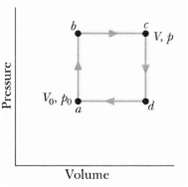
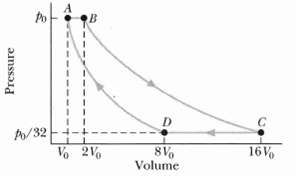
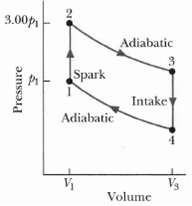
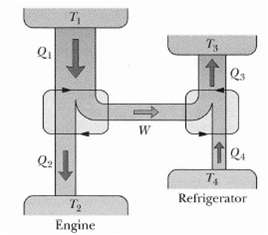
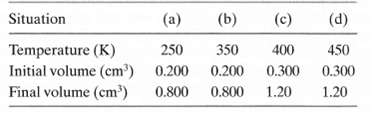
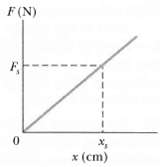
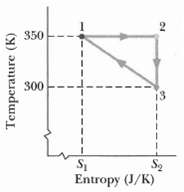
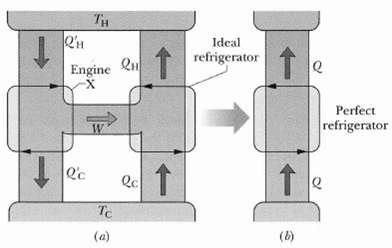
Fundamentals of Ethics for Scientists and Engineers 1st Edition Edmund G. Seebauer, Robert L. Barry - Solutions
Discover the ultimate resource for mastering "Fundamentals of Ethics for Scientists and Engineers" by Edmund G. Seebauer and Robert L. Barry. Our platform offers comprehensive online solutions, including an answers key, solution manual, and solutions PDF, ensuring all your questions and answers are covered. Dive into solved problems with step-by-step answers, perfect for preparing with our test bank and chapter solutions. Enhance your learning experience with our instructor manual and textbook resources, all available for free download. Unlock the full potential of your textbook with our expertly crafted content.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()