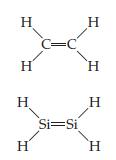
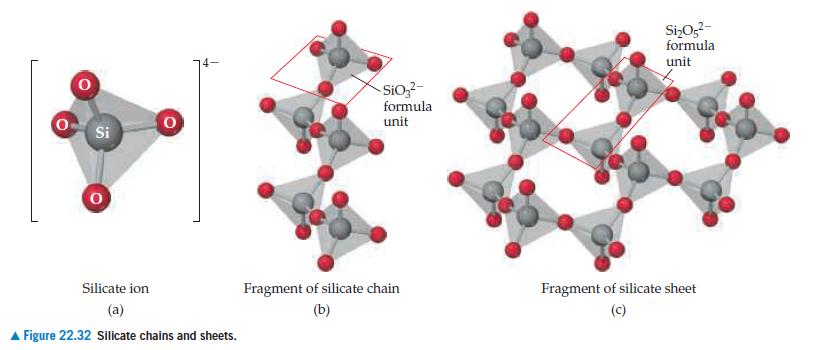
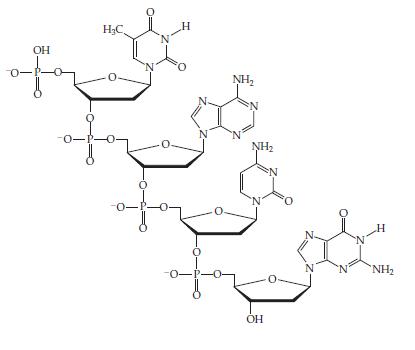
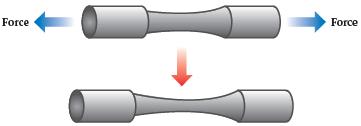
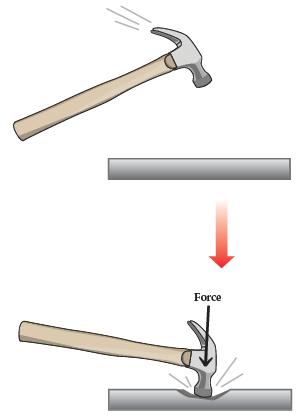
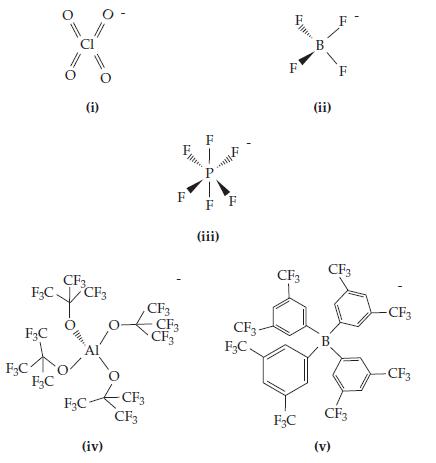
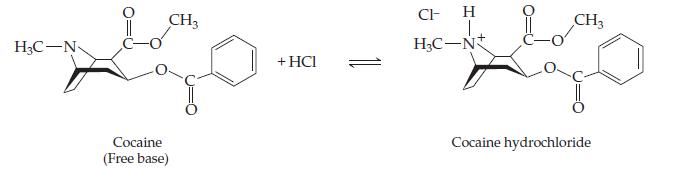
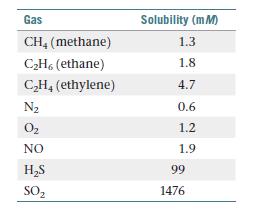
Chemistry The Central Science 14th Edition Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus - Solutions
Unlock the full potential of "Chemistry: The Central Science, 14th Edition" with our comprehensive solution manual. Access step-by-step answers, solved problems, and chapter solutions to enhance your understanding. Our online platform offers an extensive range of questions and answers, complete with an answers key for precise guidance. Whether you're looking for a test bank or an instructor manual, our resources provide invaluable support. Download solutions in PDF for free and master your textbook with ease. Perfect for students seeking detailed, reliable solutions and instructors needing a robust teaching aid.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()