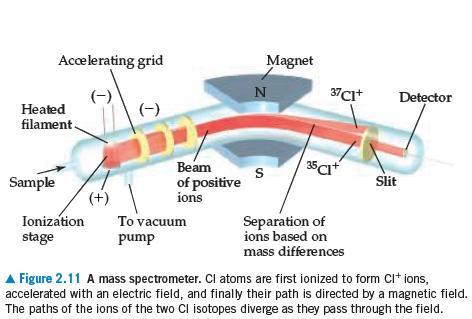
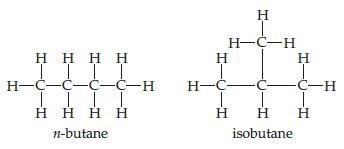
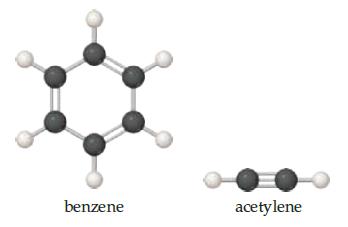
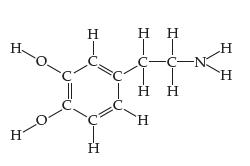
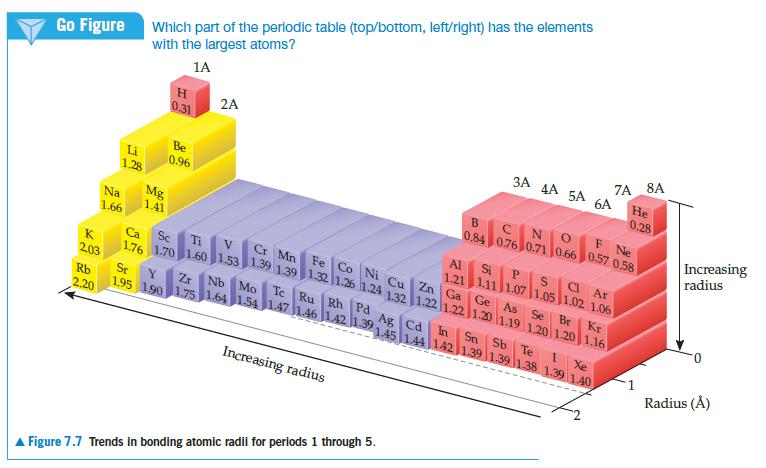
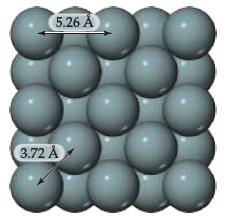
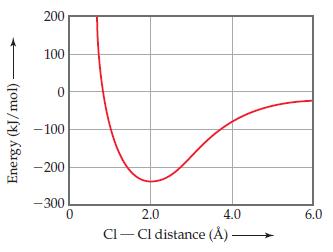
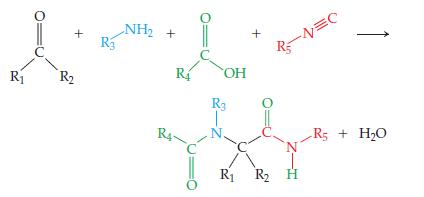
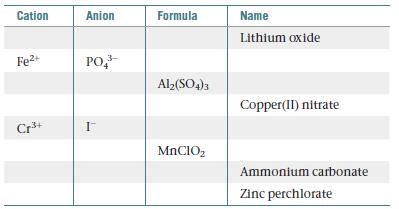
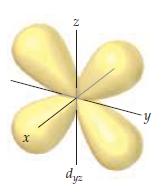
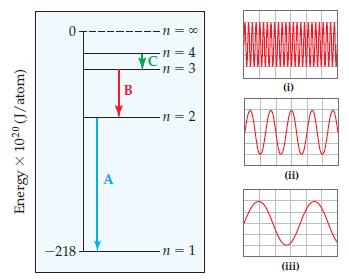
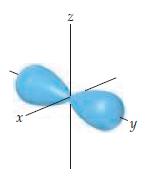
Chemistry The Central Science 13th Edition Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus - Solutions
Discover comprehensive solutions for "Chemistry: The Central Science, 13th Edition" by Theodore E. Brown and team. Access an extensive online answer key, providing detailed answers and a solution manual for better understanding. Explore chapter solutions with step-by-step answers, ideal for tackling solved problems efficiently. Benefit from a complete test bank and instructor manual designed to enhance your learning experience. This textbook resource includes questions and answers, ensuring you grasp complex concepts with ease. Download the solutions PDF for free today and transform your chemistry studies with these invaluable tools.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()