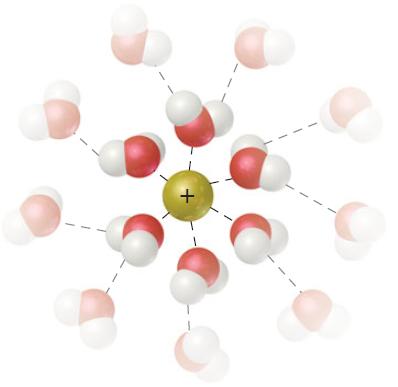
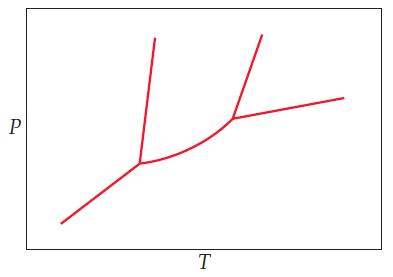
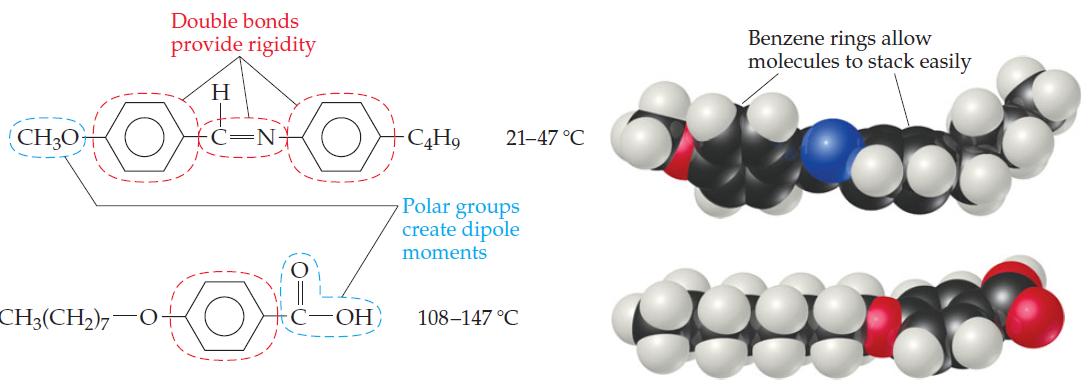
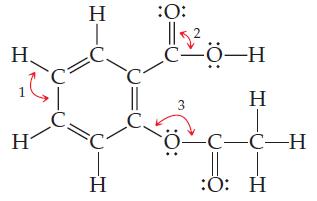
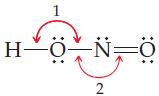
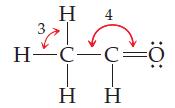
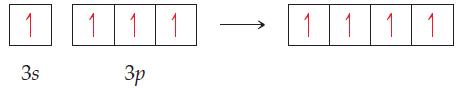
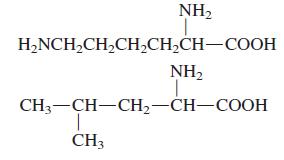
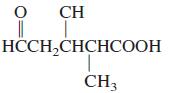
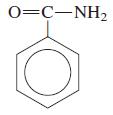
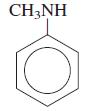
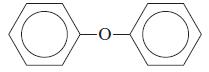
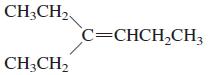
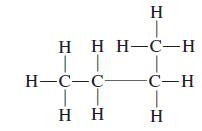
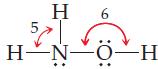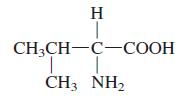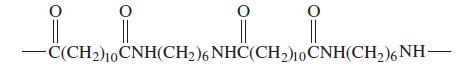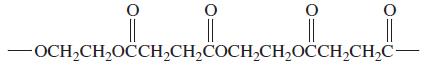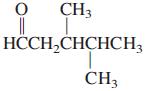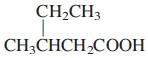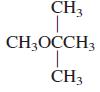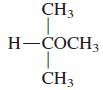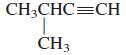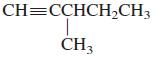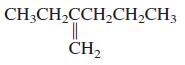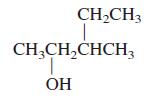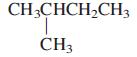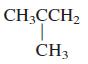General Chemistry 9th edition Darrell Ebbing, Steven D. Gammon - Solutions
Discover an extensive selection of questions and answers from "General Chemistry 9th Edition" by Darrell Ebbing and Steven D. Gammon. Our platform offers comprehensive solutions, including an instructor manual and test bank, tailored for both students and educators. Access step-by-step answers and a complete solution manual to enhance your learning experience. Whether you're looking for solved problems, chapter solutions, or the answers key, our resources are available for free download. Explore the textbook's insights with our online solutions PDF, providing a thorough understanding of chemistry concepts.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()