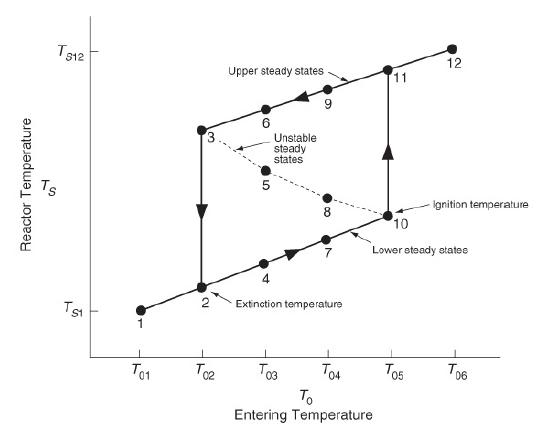
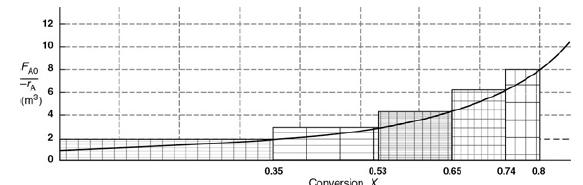
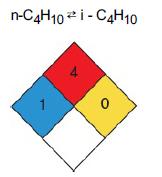
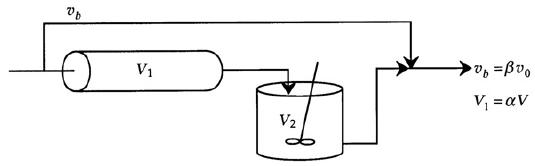
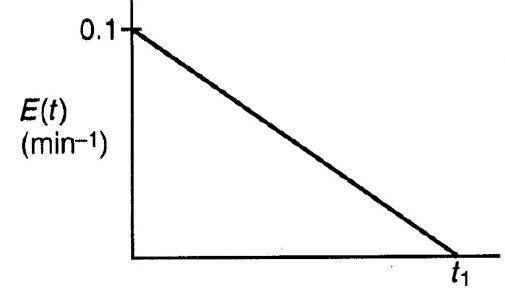
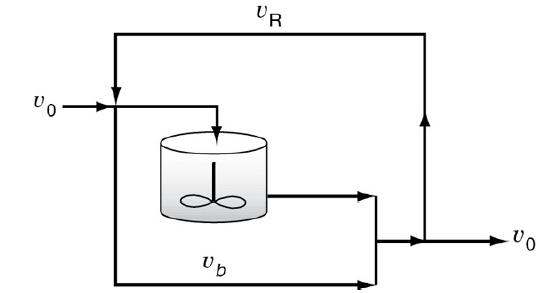
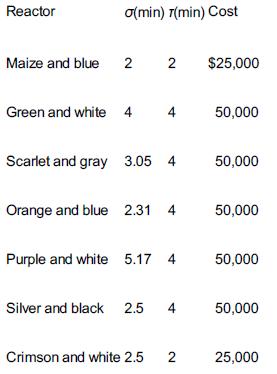
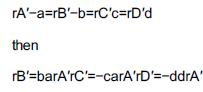Chemical Reaction Engineering 3rd Edition Octave Levenspiel - Solutions
Discover comprehensive solutions for "Chemical Reaction Engineering 3rd Edition" by Octave Levenspiel with our online resources. Access the answers key and explore detailed solutions in our solution manual, available as a free download. Dive into the solved problems and find step-by-step answers to enhance your understanding. Our questions and answers section, along with the test bank, offers valuable insights into chapter solutions. Whether you're using the instructor manual or the textbook, our solutions PDF provides an essential guide for mastering the material.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()