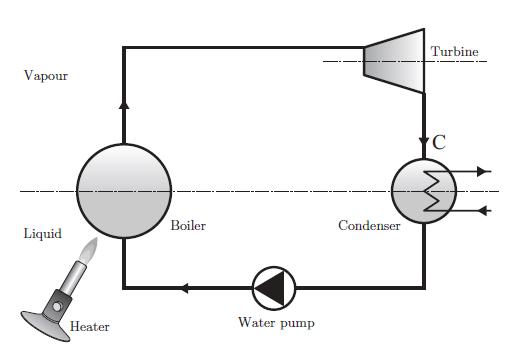
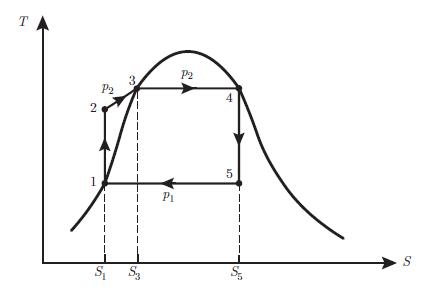
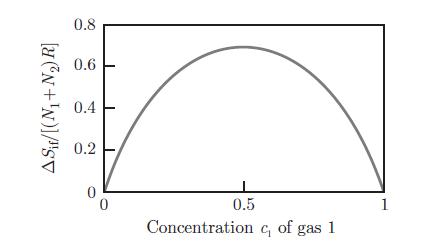
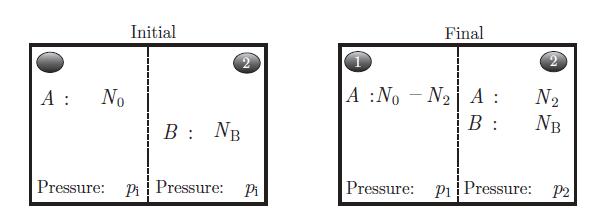
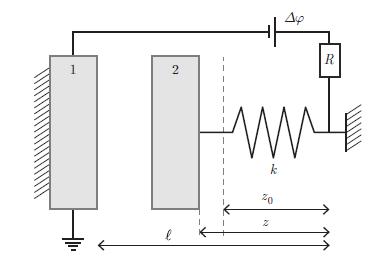
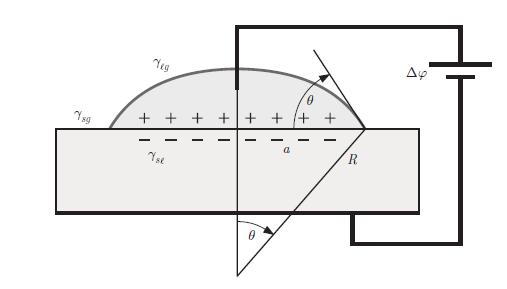
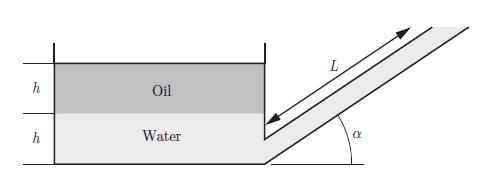
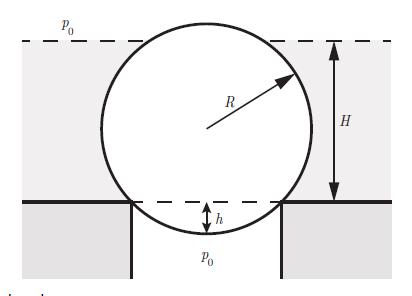
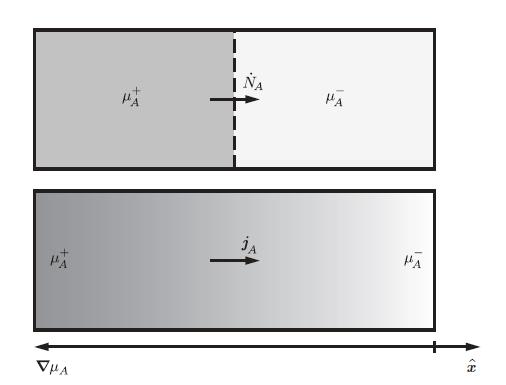
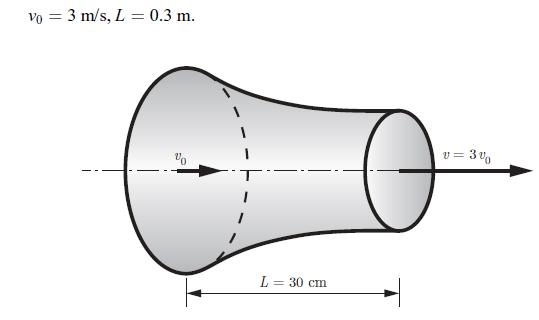
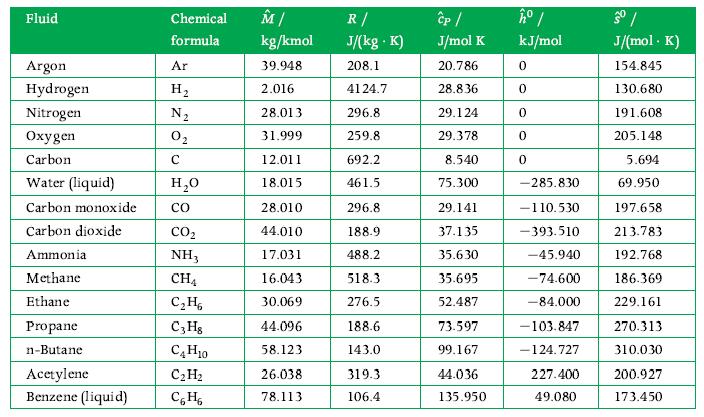
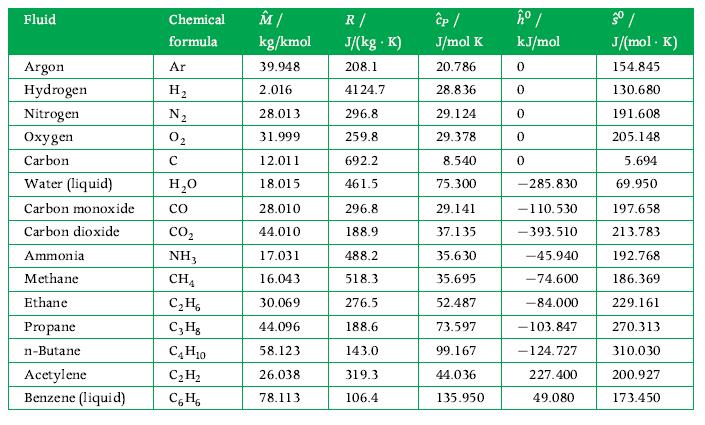
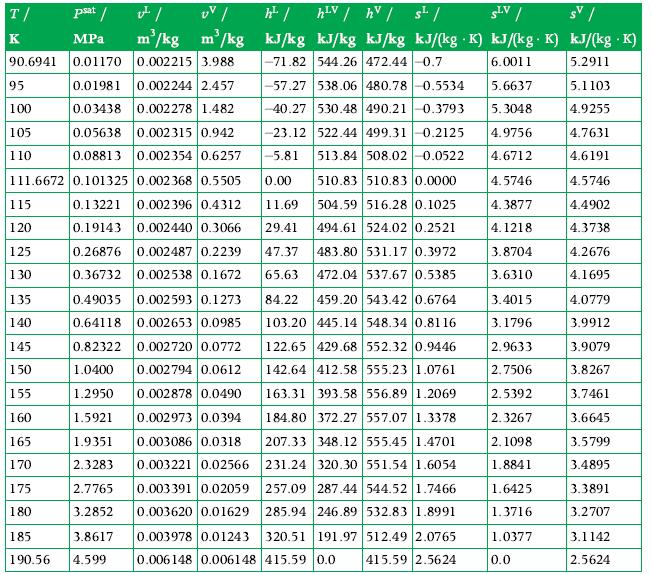
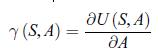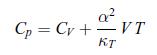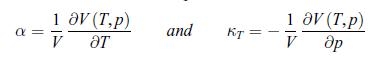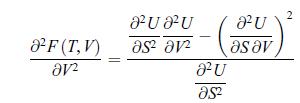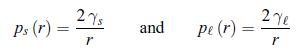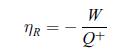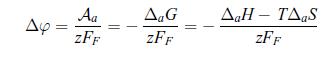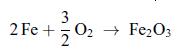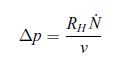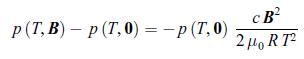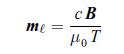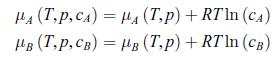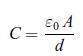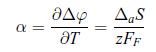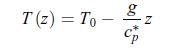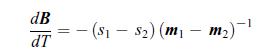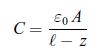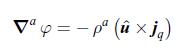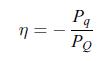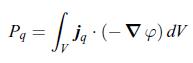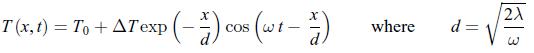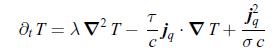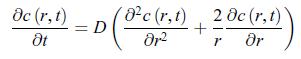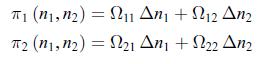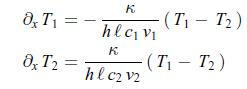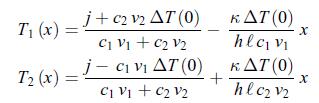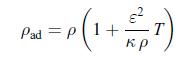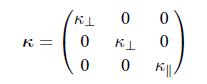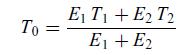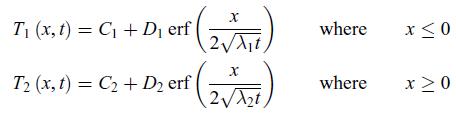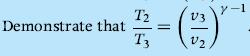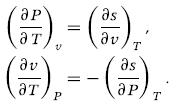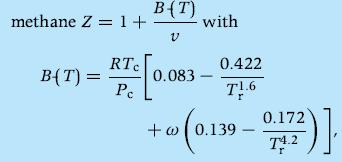Thermodynamics Fundamentals And Engineering Applications 1st Edition William C. Reynolds, Piero Colonna - Solutions
Unlock the full potential of your understanding with our comprehensive solutions for "Thermodynamics Fundamentals And Engineering Applications 1st Edition" by William C. Reynolds and Piero Colonna. Access a wide range of resources including a solution manual, solved problems, and chapter solutions available in a convenient PDF format. Whether you're seeking step-by-step answers, an instructor manual, or a test bank, our online platform provides everything you need. Enhance your learning with our textbook answers key, offering free downloads for seamless access. Dive into questions and answers with ease and elevate your thermodynamics expertise today!
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()