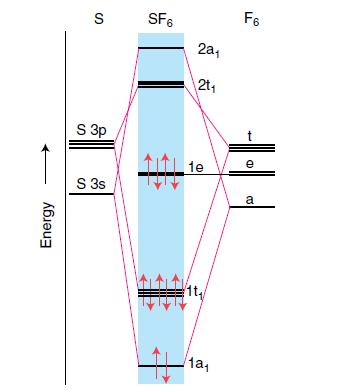
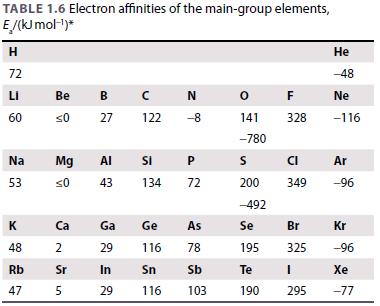
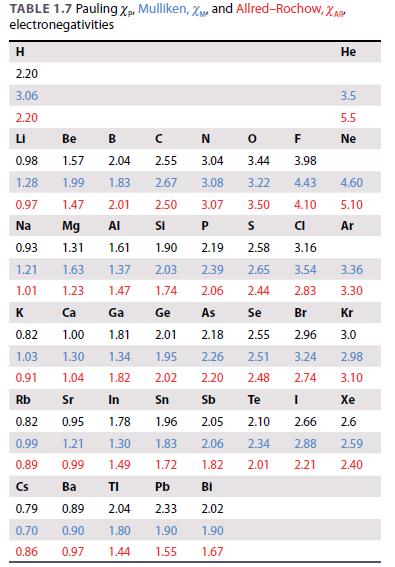
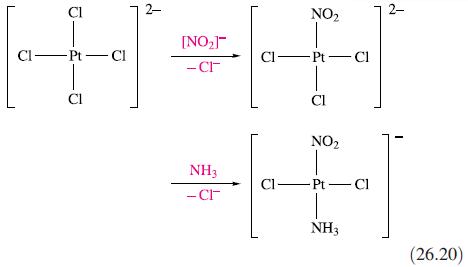
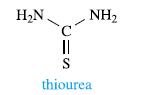
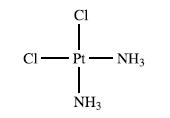
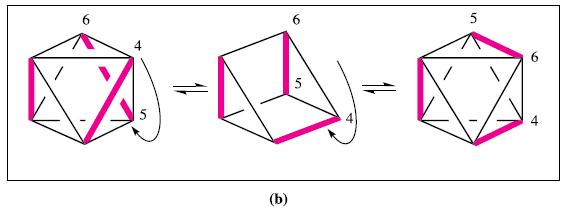
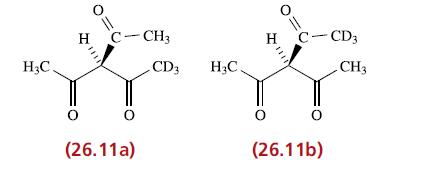
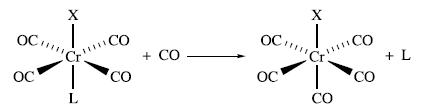
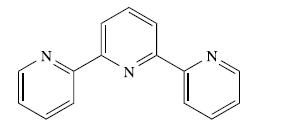
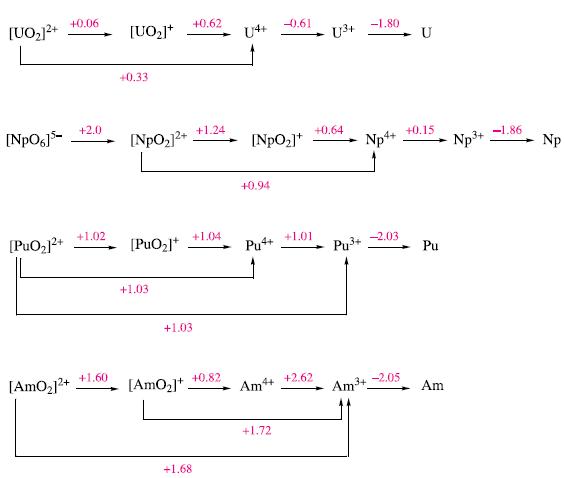
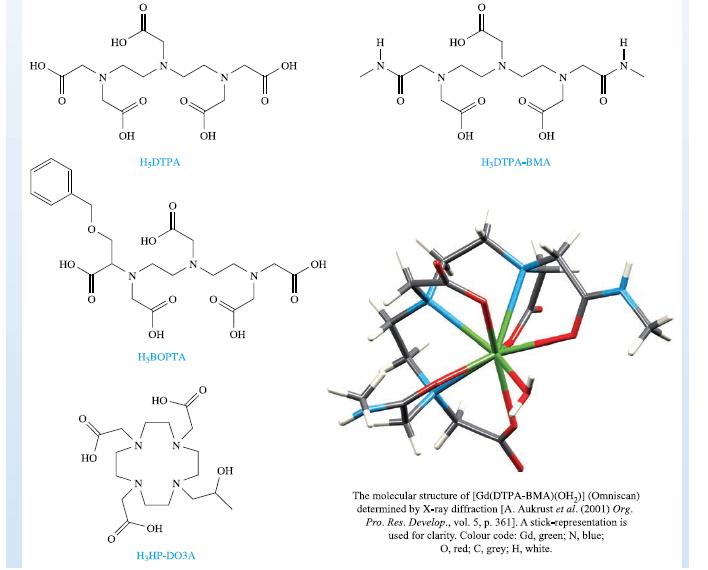
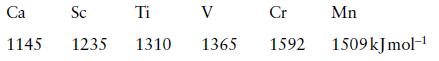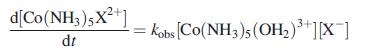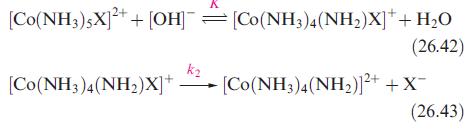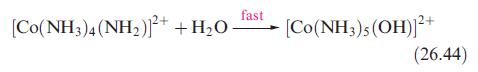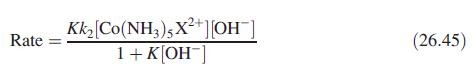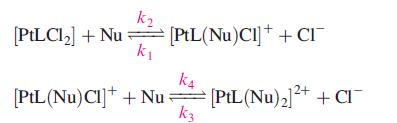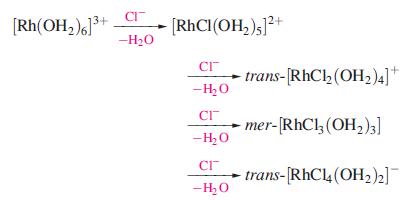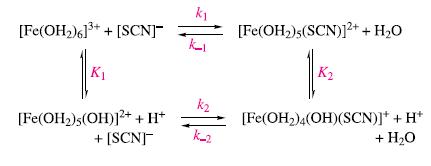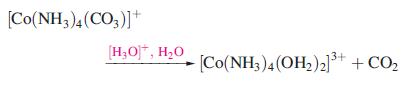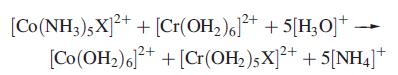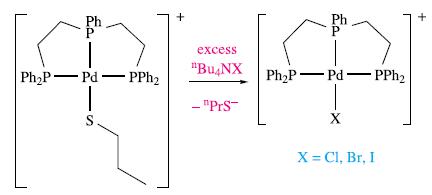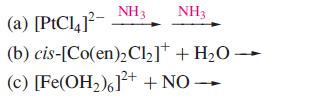Inorganic Chemistry 5th Edition Catherine Housecroft - Solutions
Enhance your study experience with our comprehensive solutions for "Inorganic Chemistry 5th Edition" by Catherine Housecroft. Access our online answers key and solutions manual to tackle even the most challenging questions. Explore detailed solutions in PDF format, featuring solved problems, questions, and answers that cater to your academic needs. Dive into our test bank and chapter solutions for a thorough understanding of each topic. Benefit from step-by-step answers guided by our instructor manual, tailored specifically for this textbook. Avail yourself of a free download to ensure you have all the resources you need for success.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()